Li-ion batteries used in automobiles typically use a LiMn2O4 cathode in place of the LiCoO2 cathode found in most Li-ion batteries. (b) Which material has a higher percentage of lithium? Does this help to explain why batteries made with LiMn2O4 cathodes deliver less power on discharging?

(b) Can the “fuel” of a fuel cell be a solid?
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
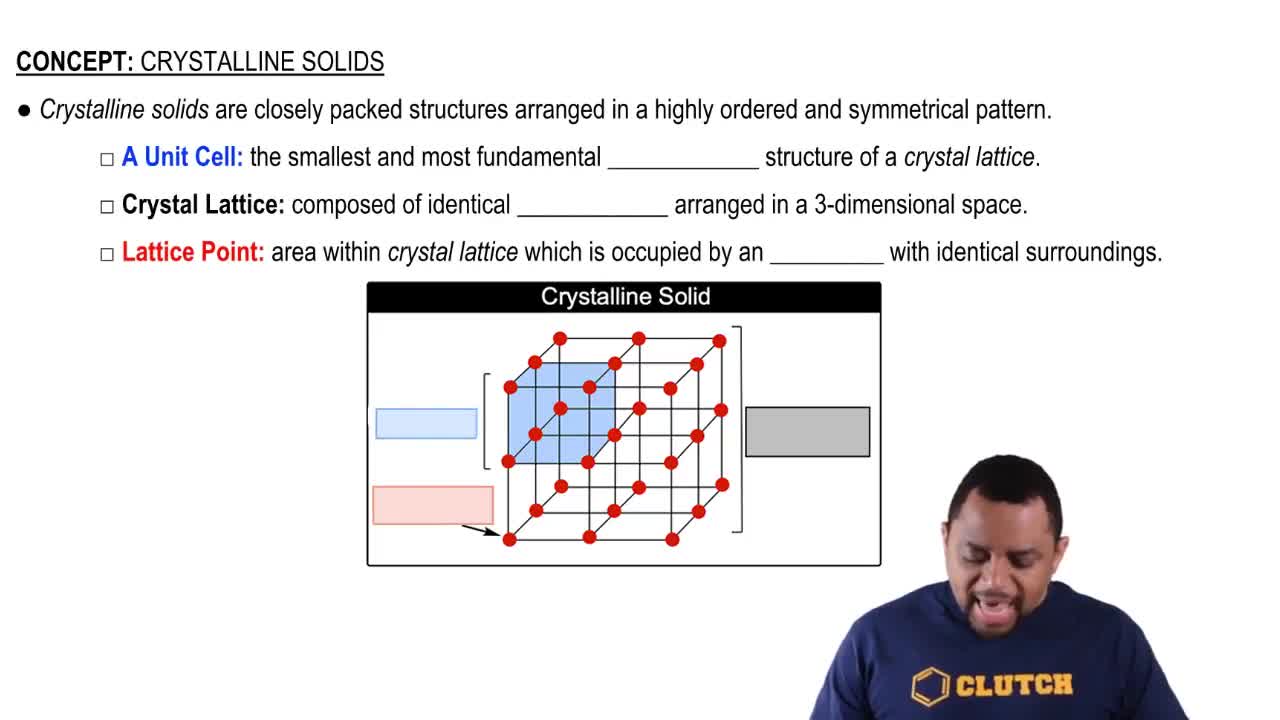
Key Concepts
Fuel Cells

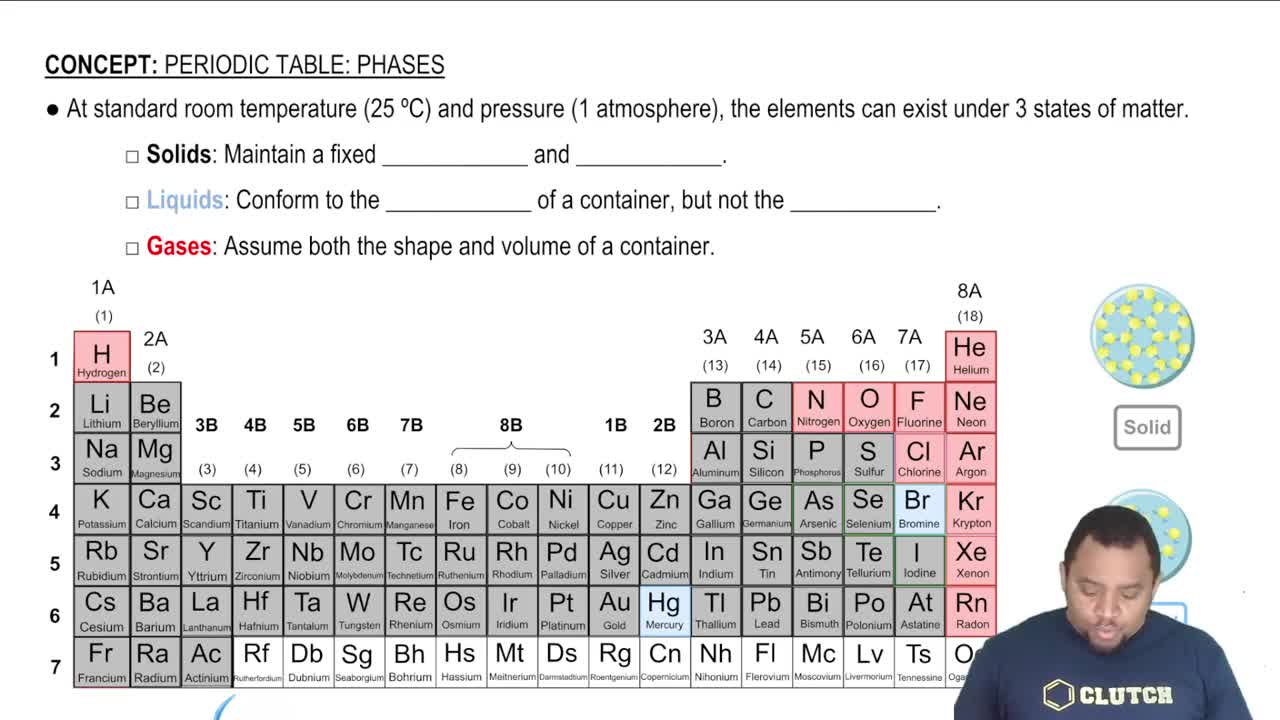
States of Matter
Solid Fuels in Energy Systems
(a) Which reaction is spontaneous in the hydrogen fuel cell: hydrogen gas plus oxygen gas makes water, or water makes hydrogen gas plus oxygen gas?
Iron corrodes to produce rust, Fe2O3, but other corrosion products that can form are Fe(O)(OH), iron oxyhydroxide, and magnetite, Fe3O4. (a) What is the oxidation number of Fe in iron oxyhydroxide, assuming oxygen's oxidation number is -2? (b) The oxidation number for Fe in magnetite was controversial for a long time. If we assume that oxygen’s oxidation number is - 2, and Fe has a unique oxidation number, what is the oxidation number for Fe in magnetite? (O)(OH), iron oxyhydroxide, and magnetite, Fe3O4. (c) It turns out that there are two different kinds of Fe in magnetite that have different oxidation numbers. Suggest what these oxidation numbers are and what their relative stoichiometry must be, assuming oxygen’s oxidation number is -2.
