Textbook Question
Write balanced equations for the following reactions. a. barium oxide with water

 Verified step by step guidance
Verified step by step guidance

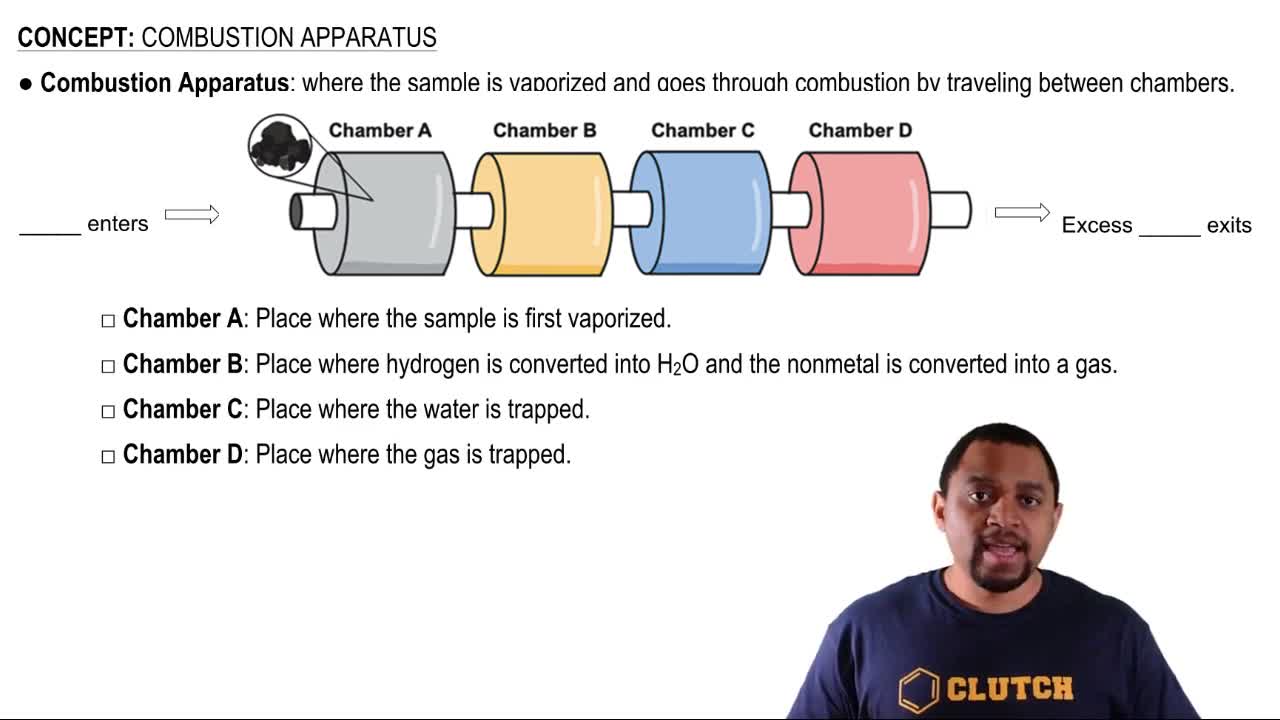
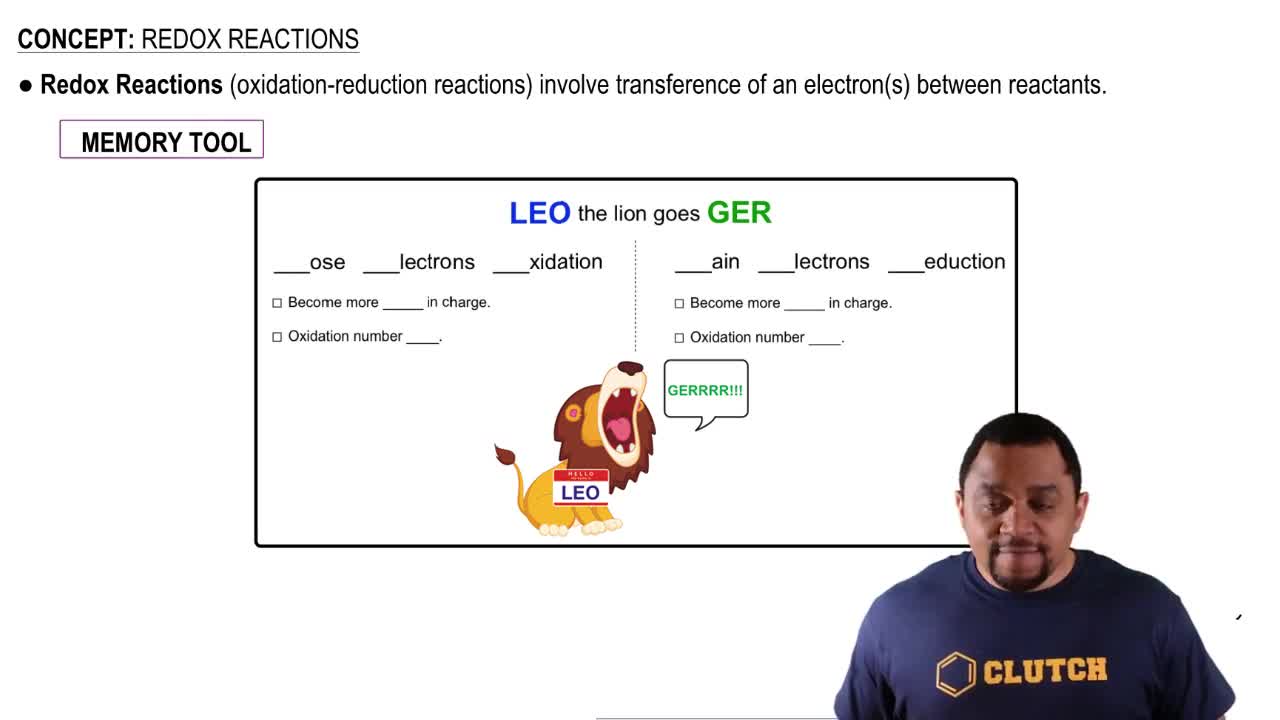
Write balanced equations for the following reactions. a. barium oxide with water
Write balanced equations for the following reactions. c. sulfur trioxide with water
Write balanced equations for the following reactions. d. selenium dioxide with aqueous potassium hydroxide.
Write a balanced equation for the reaction that occurs in each of the following cases: (a) Potassium metal is exposed to an atmosphere of chlorine gas.
Write a balanced equation for the reaction that occurs in each of the following cases: (a) Cesium is added to water.
Write a balanced equation for the reaction that occurs in each of the following cases: (c) Sodium reacts with oxygen.