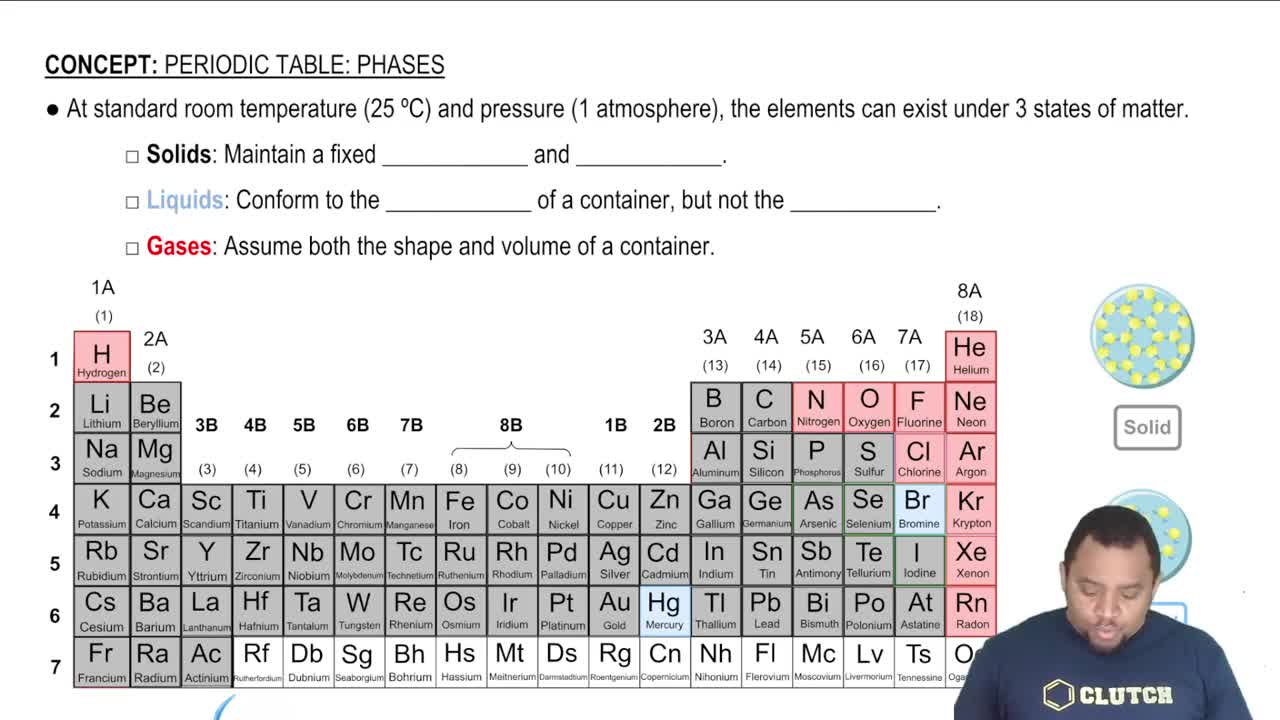
Textbook Question
Write the balanced chemical equation for the reaction of aqueous potassium hydroxide with aqueous iron(III) chloride to form solid iron(III) hydroxide and aqueous potassium chloride.
925
views

 Verified step by step guidance
Verified step by step guidance


Write the balanced chemical equation for the reaction of aqueous potassium hydroxide with aqueous iron(III) chloride to form solid iron(III) hydroxide and aqueous potassium chloride.
Write the balanced chemical equation for each reaction.
c. Sulfur dioxide gas reacts with oxygen gas to form sulfur trioxide gas.
d. Gaseous ammonia (NH3) reacts with gaseous oxygen to form gaseous nitrogen monoxide and gaseous water.
Balance each chemical equation. a. Na2S(aq) + Cu(NO3)2(aq) → NaNO3(aq) + CuS(s) b. N2H4(l) → NH3(g) + N2(g) c. HCl(aq) + O2(g) → H2O(l) + Cl2(g) d. FeS(s) + HCl(aq) → FeCl2(aq) + H2S(g)