Textbook Question
Indicate which compounds contain aldehyde or ketone carbonyl groups.
c. CH3CH2–O–CH2–CHO
813
views

 Verified step by step guidance
Verified step by step guidance
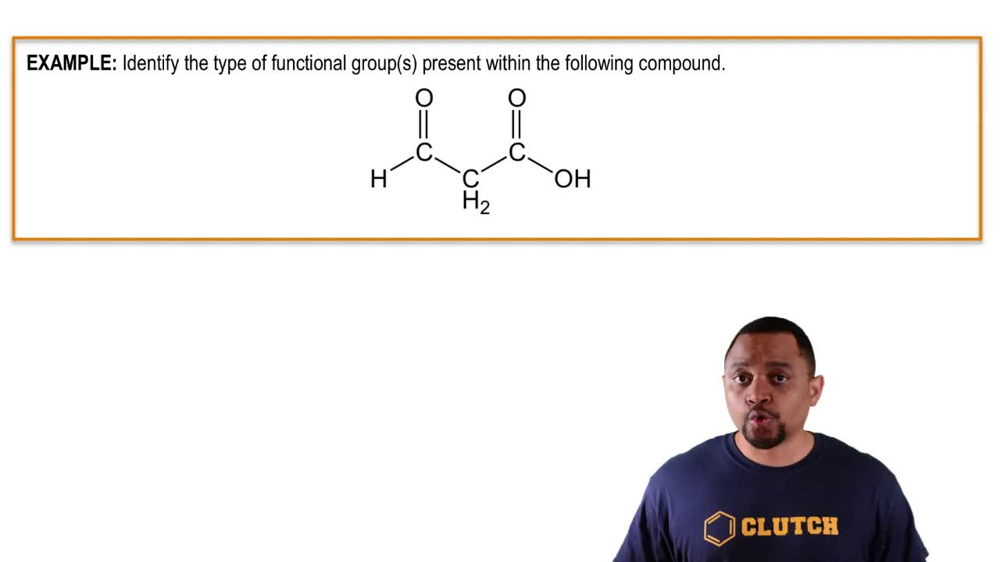


Indicate which compounds contain aldehyde or ketone carbonyl groups.
c. CH3CH2–O–CH2–CHO
Indicate which compounds contain aldehyde or ketone carbonyl groups.
f.
Redraw each of the following in line structure format. Indicate which compounds have an aldehyde carbonyl group, a ketone carbonyl group, or neither.
c.
Draw structures corresponding to the following aldehyde and ketone names:
a. 3-Methylpentanal
Draw structures corresponding to the following aldehyde and ketone names:
b. 4-Ethyl-2-isopropylhexanal
Draw structures corresponding to the following aldehyde and ketone names:
e. 1,1,1-Trichloro-3-pentanone