Textbook Question
The principal short-term energy storage molecule in cells is __________ .
793
views

 Verified step by step guidance
Verified step by step guidance


The principal short-term energy storage molecule in cells is __________ .
Which of the following structures have no electrical charge?
a. Protons
b. Electrons
c. Neutrons
d. Ions
The atomic mass of an atom most closely approximates the sum of the masses of all its __________.
a. protons
b. isotopes
c. electrons
d protons and neutrons
Name five properties of water that are vital to life.
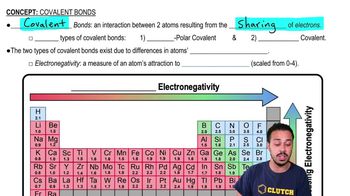
List three main types of chemical bonds, and give an example of each.
Shown is the amino acid tryptophan. Put the letter “C” at the site of every carbon atom. Label the amino group, the carboxyl group, and the side group.