What product do you expect to obtain from each of the following reactions?
a.

 Bruice 8th Edition
Bruice 8th Edition Ch. 15 - Reactions of Carboxylic Acids and Carboxylic Acid Derivatives
Ch. 15 - Reactions of Carboxylic Acids and Carboxylic Acid Derivatives Problem 89b
Problem 89b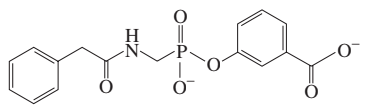
 Verified step by step guidance
Verified step by step guidance

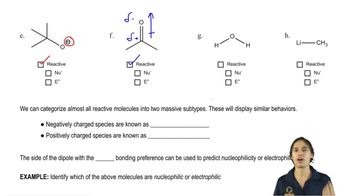
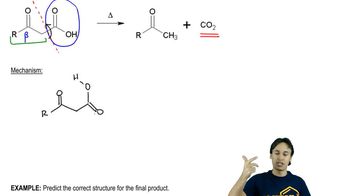
What product do you expect to obtain from each of the following reactions?
a.
Catalytic antibodies catalyze a reaction by forcing the conformation of the substrate in the direction of the transition state. The synthesis of the antibody is carried out in the presence of a transition state analog—a stable molecule that structurally resembles the transition state. This causes an antibody to be generated that recognizes and binds to the transition state, thereby stabilizing it. For example, the following transition state analog has been used to generate a catalytic antibody that catalyzes the hydrolysis of the structurally similar ester:
a. Draw a possible transition state for the hydrolysis reaction.
The following compound has been found to be an inhibitor of penicillinase. The enzyme can be reactivated by hydroxylamine (NH2OH). Propose a mechanism to account for the inhibition.
The intermediate shown here is formed during the hydroxide-ion-promoted hydrolysis of the ester group. Propose a mechanism for the reaction.
Propose a mechanism that accouts for the formation of the product.