Textbook Question
What is the product of the following reaction?
1001
views

 Mullins 1st Edition
Mullins 1st Edition Ch. 24 - Benzene II: Reactions Influenced by the Aromatic Ring
Ch. 24 - Benzene II: Reactions Influenced by the Aromatic Ring Problem 7
Problem 7 Verified step by step guidance
Verified step by step guidance

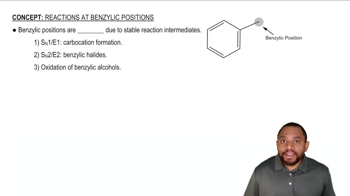

What is the product of the following reaction?
Calculate the oxidation number of the indicated atoms in the following reaction.
Rationalize the rate difference in carbocation formation for the following molecules.
Which haloalkane would you expect to undergo the fastest SN1 reaction? Why?
Though the nitro group is electron-withdrawing by resonance, when in the meta position, it doesn’t communicate by resonance with the phenoxide anion. And yet, the pKₐ value of m-nitrophenol is lowered from 10 (the pKₐ value of unsubstituted phenol) to 8.4. Explain this observation.
Which proton, Ha or Hb, would you expect to have the lower pKa value?