Provide a mechanism of the Diels–Alder reaction shown and predict the regioisomer that will form.
Ch. 22 - Conjugated Systems II: Pericyclic Reactions

Mullins1st EditionOrganic Chemistry: A Learner Centered ApproachISBN: 9780137566471Not the one you use?Change textbook
Chapter 21, Problem 32d
Predict the product of the following sigmatropic rearrangements. Be sure to rationalize the stereochemical outcome with a chair-like transition state.
(d) 
 Verified step by step guidance
Verified step by step guidance1
Identify the type of sigmatropic rearrangement involved. Sigmatropic rearrangements are pericyclic reactions where a sigma bond is shifted along with the pi system. Common examples include [1,5]- and [3,3]-sigmatropic shifts.
Determine the number of atoms involved in the rearrangement. For example, a [3,3]-sigmatropic rearrangement involves a six-membered transition state, which is often chair-like.
Draw the chair-like transition state. In a chair conformation, consider the stereochemistry of substituents. Axial and equatorial positions can influence the stereochemical outcome of the product.
Analyze the stereochemistry of the starting material and how it affects the transition state. Ensure that substituents are placed in positions that minimize steric hindrance and maximize stability.
Predict the product by completing the rearrangement in the chair-like transition state. Consider the stereochemical configuration of the product based on the positions of substituents in the transition state.

Verified video answer for a similar problem:
This video solution was recommended by our tutors as helpful for the problem above.
Video duration:
14mWas this helpful?
Key Concepts
Here are the essential concepts you must grasp in order to answer the question correctly.
Sigmatropic Rearrangements
Sigmatropic rearrangements are a type of pericyclic reaction where a sigma bond adjacent to one or more pi systems migrates across the molecule. These reactions are classified by the number of atoms involved in the migration, denoted as [i,j]-shifts. Understanding the nature of these shifts is crucial for predicting the structural changes in the product.
Recommended video:
Guided course
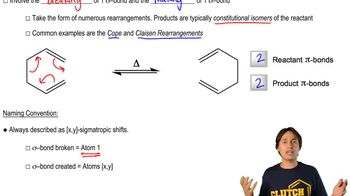
Nomenclature of Sigmatropic Shifts
Chair-like Transition State
A chair-like transition state is a conformation that resembles the chair form of cyclohexane, often adopted during pericyclic reactions to minimize steric strain and maximize orbital overlap. This conformation is particularly relevant in sigmatropic rearrangements, as it helps rationalize the stereochemical outcome by providing a stable, low-energy pathway for the reaction.
Recommended video:
Guided course

Intermediates vs. Transition States
Stereochemistry in Pericyclic Reactions
Stereochemistry in pericyclic reactions is governed by the conservation of orbital symmetry, as described by the Woodward-Hoffmann rules. These rules predict the stereochemical outcome based on the symmetry of the molecular orbitals involved. In sigmatropic rearrangements, understanding how the transition state influences stereochemistry is essential for predicting the configuration of the product.
Recommended video:
Guided course

Properties and Types of Pericyclic Reactions
Related Practice
Textbook Question
1137
views
Textbook Question
Predict the product of the following sigmatropic rearrangements. Be sure to rationalize the stereochemical outcome with a chair-like transition state.
(b)
1302
views
Textbook Question
Identify the following dienes as being in the s-cis or s-trans conformation. If they are in the s-trans conformation, draw them in the s-cis conformation. [It may not always be possible.]
(e)
1163
views
Textbook Question
Predict the product of the following electrocyclic reactions.
(c)
1022
views
Textbook Question
Identify the following dienes as being in the s-cis or s-trans conformation. If they are in the s-trans conformation, draw them in the s-cis conformation. [It may not always be possible.]
(c)
1232
views
Textbook Question
Predict the product of the following electrocyclic reactions.
(b)
508
views
