Predict the products of the sulfuric acid-catalyzed dehydration of the following alcohols. When more than one product is expected, label the major and minor products.
(d) 1-isopropylcyclohexanol
(e) 2-methylcyclohexanol

 Verified step by step guidance
Verified step by step guidance


Predict the products of the sulfuric acid-catalyzed dehydration of the following alcohols. When more than one product is expected, label the major and minor products.
(d) 1-isopropylcyclohexanol
(e) 2-methylcyclohexanol
Some alcohols undergo rearrangement or other unwanted side reactions when they dehydrate in acid. Alcohols may be dehydrated under mildly basic conditions using phosphorus oxy-chloride (POCl3) in pyridine. The alcohol reacts with phosphorus oxychloride much like it reacts with tosyl chloride (Section 11-5), displacing a chloride ion from phosphorus to give an alkyl dichlorophosphate ester. The dichlorophosphate group is an outstanding leaving group. Pyridine reacts as a base with the dichlorophosphate ester to give an E2 elimination. Propose a mechanism for the dehydration of cyclohexanol by POCl3 in pyridine.
Write balanced equations for the three preceding reactions.
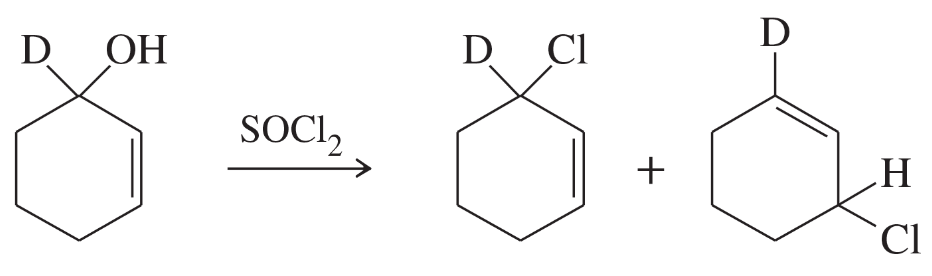
Give the structures of the products you would expect when each alcohol reacts with (1) HCl, ZnCl2; (2) HBr; (3) PBr3; (4) P/I2; and (5) SOCl2.
(a) butan-1-ol
(b) 2-methylbutan-2-ol
When cis-2-methylcyclohexanol reacts with the Lucas reagent, the major product is 1-chloro-1-methylcyclohexane. Propose a mechanism to explain the formation of this product.
Suggest how you would convert trans-4-methylcyclohexanol to
a. trans-1-chloro-4-methylcyclohexane.
b. cis-1-chloro-4-methylcyclohexane.