Draw all of the possible spin states to explain why a hydrogen with four neighbors appears as a quintet (quint, five peaks).

 Mullins 1st Edition
Mullins 1st Edition Ch. 15 - Structural Identification II: Nuclear Magnetic Resonance Spectroscopy
Ch. 15 - Structural Identification II: Nuclear Magnetic Resonance Spectroscopy Problem 27
Problem 27An unknown compound (C₇H₆O) gives the IR spectrum shown here. At what chemical shifts would you expect to see signals in the ¹H NMR spectrum?
<IMAGE>
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Infrared (IR) Spectroscopy
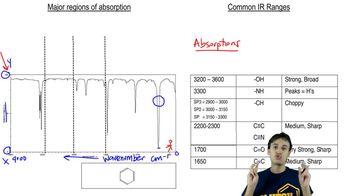
¹H Nuclear Magnetic Resonance (NMR) Spectroscopy

Aromatic Compounds

The methyl hydrogens in propane appear at a chemical shift of 0.9 ppm, whereas the methyl hydrogens of propene appear around 2.5 ppm. Explain.
(a) Calculate the resonance frequency of an aldehydic proton ( δ 9.3 ppm) if it is detected on a 60-MHz NMR spectrometer.
(b) What if it were detected on a 300-MHz instrument?
In Figure 15.34, Ha was assumed to be 'up.' How does the analysis change if we assume instead that Ha is down?
Without worrying about the relative location of the signals (i.e., the chemical shift) or the splitting patterns, draw a spectrum of the following molecule, being sure to indicate the integration of each peak. Label each signal based on the set of equivalent hydrogens to which it corresponds. [We expand on this question in future assessments.]
Though Figure 15.34 was concerned with the appearance of Ha, how would Hb appear in the spectrum?