Textbook Question
Predict the product of the following Stille coupling reactions.
(b)
1193
views


 Verified step by step guidance
Verified step by step guidance
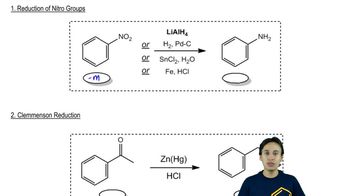


Predict the product of the following Stille coupling reactions.
(b)
Predict the product of the diorganocuprate cross-coupling reactions shown.
(b)
Predict the product of the following Sonogashira coupling reactions.
(b)
Predict the product of the diorganocuprate cross-coupling reactions shown.
(a)
Predict the product of the following Sonogashira coupling reactions.
(a)
Predict the product of the diorganocuprate cross-coupling reactions shown.
(c)