Though it wasn't discussed, what coupling constant would you expect for Hₐ and H꜀ in the spectrum of trans-but-2-enoic acid in Figure 15.50? Justify your answer.
<IMAGE>

 Mullins 1st Edition
Mullins 1st Edition Ch. 15 - Structural Identification II: Nuclear Magnetic Resonance Spectroscopy
Ch. 15 - Structural Identification II: Nuclear Magnetic Resonance Spectroscopy Problem 44
Problem 44
 Verified step by step guidance
Verified step by step guidance
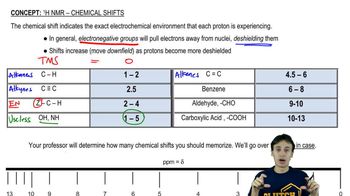

Though it wasn't discussed, what coupling constant would you expect for Hₐ and H꜀ in the spectrum of trans-but-2-enoic acid in Figure 15.50? Justify your answer.
<IMAGE>
Sketch the signals you would expect to see for Hb in the molecule shown. The important coupling constants are given.
Replace Hₐ, H₆, and H꜀ in methyl benzene with a deuterium. What is the relationship between the three products you obtain? Based on this, how many signals would you expect for these hydrogens in the ¹H NMR spectrum?
Replace Hₐ and H₆ in hexane with a deuterium. What is the relationship between the two products you obtain? Based on this, would you expect the two hydrogens to give one or two signals in the ¹H NMR spectrum?
Sketch the signals you would expect to see for H꜀ in the molecule shown. The important coupling constants are given.
Sketch the signals you would expect to see for Hₐ in the molecule shown. The important coupling constants are given.