For each structure,
1. star (*) any asymmetric carbon atoms.
2. label each asymmetric carbon as (R) or (S).
3. draw any internal mirror planes of symmetry.
4. label the structure as chiral or achiral.
5. label any meso structures.
(f)

 Verified step by step guidance
Verified step by step guidance


For each structure,
1. star (*) any asymmetric carbon atoms.
2. label each asymmetric carbon as (R) or (S).
3. draw any internal mirror planes of symmetry.
4. label the structure as chiral or achiral.
5. label any meso structures.
(f)
Convert the following perspective formulas to Fischer projections.
(a)
(b)
For each structure,
1. star (*) any asymmetric carbon atoms.
2. label each asymmetric carbon as (R) or (S).
3. draw any internal mirror planes of symmetry.
4. label the structure as chiral or achiral.
5. label any meso structures.
(e)
For each structure,
1. star (*) any asymmetric carbon atoms.
2. label each asymmetric carbon as (R) or (S).
3. draw any internal mirror planes of symmetry.
4. label the structure as chiral or achiral.
5. label any meso structures.
(g)
(h)
Convert the following Fischer projections to perspective formulas.
(a)
(b)
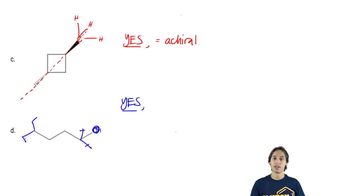
For each of the compounds described by the following names,
1. draw a three-dimensional representation.
2. star (*) each chiral center.
3. draw any planes of symmetry.
4. draw any enantiomer.
5. draw any diastereomers.
6. label each structure you have drawn as chiral or achiral.
c. (2R,3S)-2,3-dibromohexane
d. (1R,2R)-1,2-dibromocyclohexane