Rank the following compounds in decreasing order of their reactivity toward the SN2 reaction with sodium ethoxide (Na+ –OCH2CH3) in ethanol.
methyl chloride
tert-butyl iodide
neopentyl bromide
isopropyl bromide
methyl iodide
ethyl chloride

 Verified step by step guidance
Verified step by step guidance
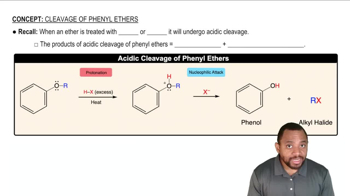

Rank the following compounds in decreasing order of their reactivity toward the SN2 reaction with sodium ethoxide (Na+ –OCH2CH3) in ethanol.
methyl chloride
tert-butyl iodide
neopentyl bromide
isopropyl bromide
methyl iodide
ethyl chloride
For each pair of compounds, state which compound is the better SN2 substrate.
a. 2-methyl-1-iodopropane or tert-butyl iodide.
b. cyclohexyl bromide or 1-bromo-1-methylcyclohexane
For each pair, predict the stronger nucleophile in the SN2 reaction (using an alcohol as the solvent). Explain your prediction.
c. NH3 or PH3
d. CH3S– or H2S
For each pair, predict the stronger nucleophile in the SN2 reaction (using an alcohol as the solvent). Explain your prediction.
e. (CH3)3N or (CH3)2O
f. CH3COO– or CF3COO–
For each pair of compounds, state which compound is the better SN2 substrate.
c. 2-bromobutane or isopropyl bromide
d. 1-chloro-2,2-dimethylbutane or 2-chlorobutane
e. 1-iodobutane or 2-iodopropane
For each pair, predict the stronger nucleophile in the SN2 reaction (using an alcohol as the solvent). Explain your prediction.
g. (CH3)2CHO– or CH3CH2CH2O–
h. I– or Cl–