Show how you would accomplish the following syntheses efficiently (you may use any necessary reagents).
(a) trans-1-bromobut-2-ene → trans-pent-3-enoic acid (two ways)

 Verified step by step guidance
Verified step by step guidance

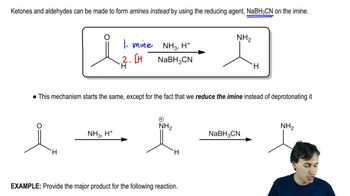

Show how you would accomplish the following syntheses efficiently (you may use any necessary reagents).
(a) trans-1-bromobut-2-ene → trans-pent-3-enoic acid (two ways)
Show how you would accomplish the following syntheses efficiently (you may use any necessary reagents).
(f)
Show how you would accomplish the following syntheses efficiently (you may use any necessary reagents).
(g)
Show how you would accomplish the following syntheses efficiently (you may use any necessary reagents).
(b) hex-3-ene → propanoic acid
Show how you would accomplish the following syntheses efficiently (you may use any necessary reagents).
(c) but-2-enal → but-2-enoic acid
Show how you would accomplish the following syntheses efficiently (you may use any necessary reagents).
(e)