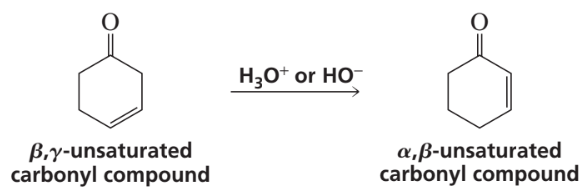
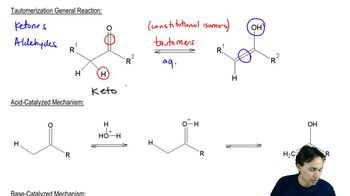
There are other condensation reactions similar to the aldol and Claisen condensations:
c. The Knoevenagel condensation is the condensation of an aldehyde or a ketone that has no a-hydrogens and a compound such as diethyl malonate that has an a-carbon flanked by two electron-withdrawing groups. Draw the product obtained from the following Knoevenagel condensation: