Propose a mechanism for reaction of the first three propylene units in the polymerization of propylene in the presence of a peroxide.

The following cyclization has been observed in the oxymercuration-demercuration of this unsaturated alcohol. Propose a mechanism for this reaction.
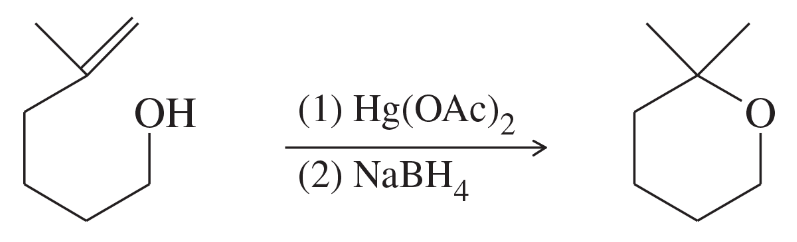
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Oxymercuration-Demercuration

Mechanism of Cyclization
Stereochemistry

An unknown compound decolorizes bromine in carbon tetrachloride, and it undergoes catalytic reduction to give decalin. When treated with warm, concentrated potassium permanganate, this compound gives cis-cyclohexane-1,2-dicarboxylic acid and oxalic acid. Propose a structure for the unknown compound.
Predict the product of formula C7H13BrO from the reaction of this same unsaturated alcohol with bromine. Propose a mechanism to support your prediction.
The two butenedioic acids are called fumaric acid (trans) and maleic acid (cis). 2,3-Dihydroxybutanedioic acid is called tartaric acid.
Show how you would convert
c. maleic acid to (±)-tartaric acid.
The two butenedioic acids are called fumaric acid (trans) and maleic acid (cis). 2,3-Dihydroxybutanedioic acid is called tartaric acid.
Show how you would convert
b. fumaric acid to meso-tartaric acid.
A graduate student attempted to form the iodohydrin of the alkene shown below. Her analysis of the products showed a good yield of an unexpected product. Propose a mechanism to explain the formation of this product.
