Textbook Question
For each pair, choose the nucleophile that would react most quickly in an SN2 reaction (assume H2O is the solvent).
(c)
1112
views

 Mullins 1st Edition
Mullins 1st Edition Ch. 12 - Substitution and Elimination: Reactions of Haloalkanes
Ch. 12 - Substitution and Elimination: Reactions of Haloalkanes Problem 57d
Problem 57d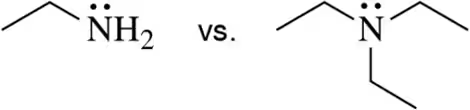
 Verified step by step guidance
Verified step by step guidance


For each pair, choose the nucleophile that would react most quickly in an SN2 reaction (assume H2O is the solvent).
(c)
For each pair, choose the nucleophile that would react most quickly in an SN2 reaction (assume H2O is the solvent).
(b)
For each pair, choose the haloalkane that would react most quickly in an SN2 reaction.
(b)
For each pair, choose the nucleophile that would react most quickly in an SN2 reaction (assume H2O is the solvent).
(a) F- vs Br-
For each pair, choose the haloalkane that would react most quickly in an SN2 reaction.
(a)
For each pair, choose the haloalkane that would react most quickly in an SN2 reaction.
(c)