Textbook Question
Predict the major products of the following reactions.
(e)
1009
views

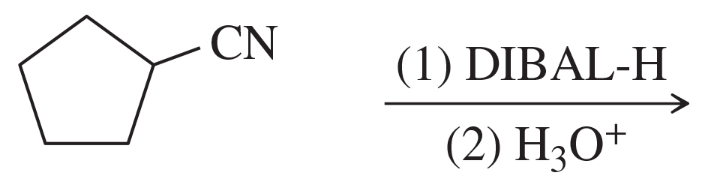
 Verified step by step guidance
Verified step by step guidance



Predict the major products of the following reactions.
(e)
Predict the major products of the following reactions.
(a)
Predict the major products of the following reactions.
(f)
Predict the major products of the following reactions.
(d)
Name the following ketones and aldehydes. When possible, give both a common name and an IUPAC name.
(l)
Predict the major products of the following reactions.
(b)