The following compound has only one asymmetric center. Why then does it have four stereoisomers?

The stereoisomer of cholesterol found in nature is shown here.
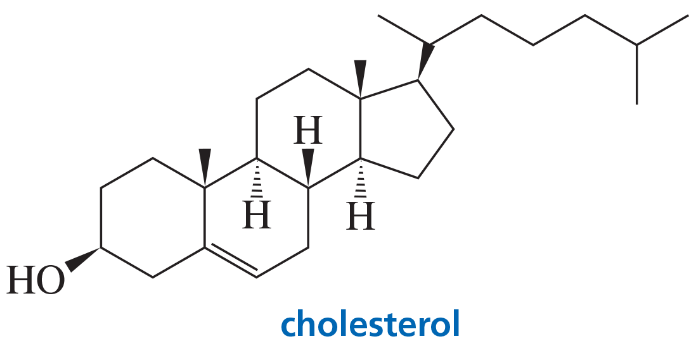
a. How many asymmetric centers does cholesterol have?
b. What is the maximum number of stereoisomers that cholesterol can have?
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Asymmetric Centers

Stereoisomers

Cholesterol Structure

Draw the stereoisomers of the following amino acids. Indicate pairs of enantiomers and pairs of diastereomers.
a.
a. Stereoisomers with two asymmetric centers are called ___ if the configuration of both asymmetric centers in one stereoisomer is the opposite of the configuration of the symmetric centers in the other stereoisomer.
b. Stereoisomers with two asymmetric centers are called ___ if the configuration of both asymmetric centers in one stereoisomer is the same as the configuration of the asymmetric centers in the other stereoisomer.
c. Stereoisomers with two asymmetric centers are called ___ if one of the asymmetric centers has the same configuration in both stereoisomers and the other asymmetric center has the opposite configuration in the two stereoisomers.
Draw all possible stereoisomers for each of the following:
b. 2-bromo-4-chlorohexane
1-Bromo-2-methylcyclopentane has four pairs of diastereomers. Draw the four pairs.
Naproxen, a nonsteroidal anti-inflammatory drug that is the active ingredient in Aleve (p. 115), has a specific rotation of +66. One commercial preparation results in a mixture with a 97% enantiomeric excess.
a. Does naproxen have the R or the S configuration?
b. What percent of each enantiomer is obtained from the commercial preparation?
