Textbook Question
Draw the ground-state electronic configuration for each of the following:
b. Ca2+
1369
views

 Bruice 8th Edition
Bruice 8th Edition Ch. 1 - Remembering General Chemistry: Electronic Structure and Bonding (Part 1)
Ch. 1 - Remembering General Chemistry: Electronic Structure and Bonding (Part 1) Problem 41
Problem 41 Verified step by step guidance
Verified step by step guidance
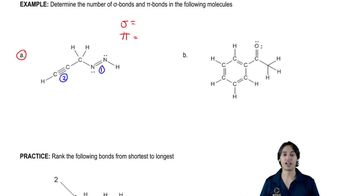


Draw the ground-state electronic configuration for each of the following:
b. Ca2+
Draw a Lewis structure for each of the following:
b. CO32−
Draw the ground-state electronic configuration for each of the following:
a. Mg
a. Which bond would be longer?
b. Which bond would be stronger?
3. H—Cl or H—F
a. Which bond would be longer?
b. Which bond would be stronger?
2. C—C or C—Cl
a. Which bond would be longer?
b. Which bond would be stronger?
1. C—Cl or C—I