Predict the theoretical number of different NMR signals produced by each compound, and give approximate chemical shifts. Point out any diastereotopic relationships.
c. Ph—CHBr—CH2Br
d. vinyl chloride

 Verified step by step guidance
Verified step by step guidance

Predict the theoretical number of different NMR signals produced by each compound, and give approximate chemical shifts. Point out any diastereotopic relationships.
c. Ph—CHBr—CH2Br
d. vinyl chloride
The spectrum of trans-hex-2-enoic acid follows.
(a) Assign peaks to show which protons give rise to which peaks in the spectrum.
(b) Draw a tree to show the complex splitting of the vinyl proton centered around 7 ppm. Estimate the values of the coupling constants.
Propose a structure that corresponds to each spectrum.
(a) <IMAGE>
Propose mechanisms to show the interchange of protons between ethanol molecules under
(a) acid catalysis.
(b) base catalysis.
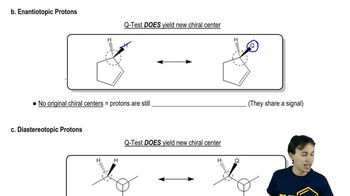
Predict the theoretical number of different NMR signals produced by each compound, and give approximate chemical shifts. Point out any diastereotopic relationships.
a. 2-bromobutane
b. cyclopentanol
Draw a splitting tree, similar to Figures 13-32 and 13-33, for proton Hc in styrene. What is the chemical shift of proton Hc?