Textbook Question
Show how you would add a Grignard reagent to an ester or a nitrile to synthesize
(c) pentan-2-one.
706
views

 Verified step by step guidance
Verified step by step guidance
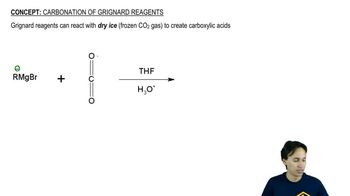


Show how you would add a Grignard reagent to an ester or a nitrile to synthesize
(c) pentan-2-one.
Draw a mechanism for the acidic hydrolysis of the magnesium salt shown below to acetophenone.
Draw a mechanism for the reaction of propanoyl chloride with 2 moles of phenylmagnesium bromide.
Show how you would add a Grignard reagent to an ester or a nitrile to synthesize
(b) heptan-4-ol.
Show how Friedel–Crafts acylation might be used to synthesize the following compounds.
a. acetophenone
Propose a mechanism for the reduction of octanoyl chloride by lithium aluminum hydride.