a. Draw the structure of cis-CH3CH=CHCH2CH3 showing the pi bond with its proper geometry.
b. Circle the six coplanar atoms in this compound.

 Verified step by step guidance
Verified step by step guidance


a. Draw the structure of cis-CH3CH=CHCH2CH3 showing the pi bond with its proper geometry.
b. Circle the six coplanar atoms in this compound.
Circle the coplanar atoms in the following structure:
Which of the following compounds show cis-trans isomerism? Draw the cis and trans isomers of the ones that do.
(d)
(e)
(f) CH3CH=NCH3
Give the relationships between the following pairs of structures. The possible relationships are as follows: same compound, cis-trans isomers, constitutional (structural) isomers, and not isomers (different molecular formula).
(a) CH3CH2CH2CH3 and (CH3)3CH
(b) CH2=CH–CH2Cl and CHCl=CH–CH3
Which of the following compounds show cis-trans isomerism? Draw the cis and trans isomers of the ones that do.
(a) CH3CH=CHCH3
(b) CH3C≡CCH3
(c) CH2=C(CH3)2
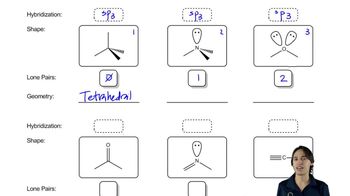
Draw orbital pictures of the pi bonding in the following compounds:
f. CH3CH=NCH=C=O