What is each compound's systematic name?
d.
e.
f.

 Bruice 8th Edition
Bruice 8th Edition Ch. 5 - Alkenes: Structure, Nomenclature, and an Introduction to Reactivity • Thermodynamics and Kinetics
Ch. 5 - Alkenes: Structure, Nomenclature, and an Introduction to Reactivity • Thermodynamics and Kinetics Problem 42d,e,f
Problem 42d,e,f Verified step by step guidance
Verified step by step guidance
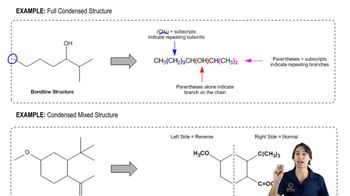

What is each compound's systematic name?
d.
e.
f.
Draw the condensed structure for each of the following:
a. (Z)-1,3,5-tribromo-2-pentene
b. (Z)-3-methyl-2-heptene
c. (E)-1,2-dibromo-3-isopropyl-2-hexene
Draw the skeletal structures for the compounds in Problem 42.
d. vinyl bromide
e. 1,2-dimethylcyclopentene
f. diallylamine
Draw the structure of a hydrocarbon that has six carbon atoms and
a. three vinylic hydrogens and two allylic hydrogens.
b. three vinylic hydrogens and one allylic hydrogen.
c. three vinylic hydrogens and no allylic hydrogens.
a. Draw the condensed structures and give the systematic names for all the alkenes with molecular formula C6H12, ignoring stereoisomers. (Hint: There are 13.)
b. Which of the alkenes have E and Z isomers?
c. Which of the alkenes is the most stable?
d. Which of the alkenes is the least stable?
Name the following:
a.