Textbook Question
Draw a skeletal structure for each of the compounds.
c. CH3COOH
1541
views

 Bruice 8th Edition
Bruice 8th Edition Ch. 1 - Remembering General Chemistry: Electronic Structure and Bonding (Part 2)
Ch. 1 - Remembering General Chemistry: Electronic Structure and Bonding (Part 2) Problem 60a,b,c
Problem 60a,b,c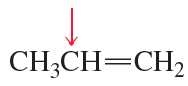
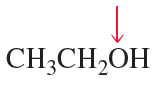
 Verified step by step guidance
Verified step by step guidance

Draw a skeletal structure for each of the compounds.
c. CH3COOH
Draw a Lewis structure for each of the following:
b. CH3OCH3
Show the direction of the dipole moment in each of the following bonds (use the electronegativities given in [Table 1.3] ):
a. H3C—Br
b. H3C—Li
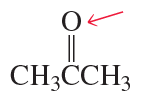
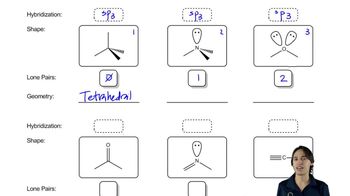
What is the hybridization of the indicated atom in each of the following?
d.
e.
f.
Predict the approximate bond angles for the following:
c. the C—C—N bond angle in CH3C≡N
d. the C—C—N bond angle in CH3CH2NH2
Draw a skeletal structure for each of the compounds.
a. CH3CHO
b. CH3OCH3