 Back
BackChemical Equilibrium, Solubility, and Acid-Base Chemistry: Analytical Chemistry Study Notes
Study Guide - Smart Notes

Chemical Equilibrium and Thermodynamics
Introduction to Chemical Equilibrium
Chemical equilibrium is a dynamic state in which the rates of the forward and reverse reactions are equal, resulting in constant concentrations of reactants and products. Understanding equilibrium is essential for predicting the composition of chemical systems and for analytical calculations.
Equilibrium Constant (K): For a general reaction , the equilibrium constant is given by:
Standard State: Solutions: 1 M, Gases: 1 bar, Pure solids/liquids: activity = 1.
Reaction Quotient (Q): Same form as K, but with current (not equilibrium) concentrations.
Direction of Reaction: If Q < K, reaction proceeds forward; if Q > K, reaction proceeds in reverse; if Q = K, system is at equilibrium.
Gibbs Free Energy and Equilibrium
The relationship between Gibbs free energy and equilibrium determines the spontaneity of reactions.
Gibbs Free Energy Change:
At Equilibrium: ,
Standard Free Energy and K:
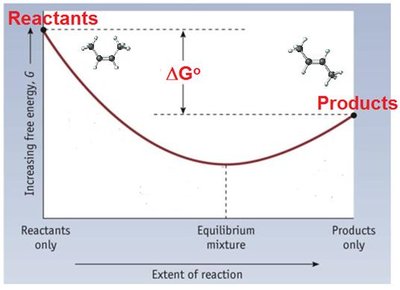
Interpretation: Negative indicates a spontaneous process; positive indicates non-spontaneity.
Solubility and Precipitation Equilibria
Solubility Product Constant (Ksp)
The solubility product constant () describes the equilibrium between a solid and its ions in solution. It is crucial for predicting precipitation and dissolution processes.
Definition: is the equilibrium constant for the dissolution of a sparingly soluble salt.
Example: For ,
Solubility: The maximum amount of solute that can dissolve in a given volume of solvent at equilibrium.

Precipitation Criteria: If , a precipitate forms; if , no precipitate forms; if , the solution is saturated.
Common Ion Effect
The presence of a common ion decreases the solubility of a salt due to Le Chatelier's Principle.
Example: Adding to a solution containing decreases its solubility.
Systematic Treatment of Equilibria
Steps for Solving Equilibrium Problems
Complex equilibrium systems are solved using a systematic approach:
Write all pertinent reactions and equilibria.
Write the charge balance equation (sum of positive charges = sum of negative charges).
Write the mass balance equation(s) (conservation of matter for each component).
Write the equilibrium constant expressions for all reactions.
Verify the number of equations matches the number of unknowns.
Solve the system of equations.
Acid-Base Equilibria
Bronsted-Lowry Acids and Bases
Acids are proton donors, and bases are proton acceptors. Acid-base equilibria are central to analytical chemistry.
Strong Acids/Bases: Completely dissociate in water (e.g., HCl, NaOH).
Weak Acids/Bases: Partially dissociate; equilibrium is important (e.g., acetic acid, ammonia).
pH and pOH: , ,
Relationship:
Buffer Solutions
Buffers resist changes in pH upon addition of small amounts of acid or base. They consist of a weak acid and its conjugate base or a weak base and its conjugate acid.
Henderson-Hasselbalch Equation:
Buffer Capacity: Greatest when and increases with higher concentrations of buffer components.
Activity and Ionic Strength
Activity Coefficients and Non-Ideal Solutions
In real solutions, ion interactions cause deviations from ideality. Activities () replace concentrations in equilibrium expressions, and activity coefficients () correct for non-ideality.
Activity:
Ionic Strength ():
Debye-Hückel and Davies Equations: Used to estimate for ions in solution.
Effect on pH Measurement
pH meters measure hydrogen ion activity, not concentration. At higher ionic strengths, activity coefficients must be considered for accurate pH calculations.
Tables
Detection Limits of Atomic Spectroscopy Methods
The following table compares the detection limit ranges for various atomic spectroscopy techniques:
Technique | Detection Limit Range (μg/L) |
|---|---|
Flame AA | ~1–100 |
ICP Emission - Radial | ~0.1–10 |
ICP Emission - Axial | ~0.01–1 |
Hydride Generation AA | ~0.01–1 |
GFAA | ~0.001–0.1 |
ICP-MS | ~0.0001–0.01 |
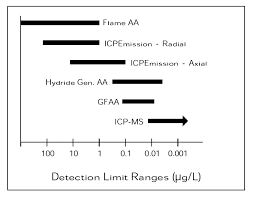
Additional info: Lower detection limits indicate higher sensitivity. ICP-MS offers the lowest detection limits among the listed techniques.
Summary Table: Standard States and Equilibrium Concentrations
State | Standard State | Equilibrium Concentration |
|---|---|---|
Solution | 1 M | CA in M |
Gas | 1 bar | PA in bar |
Liquid | Pure liquid | 1 |
Solid | Pure solid | 1 |
Key Equations
Equilibrium Constant:
Reaction Quotient:
Gibbs Free Energy:
Relationship between and :
Solubility Product:
Henderson-Hasselbalch:
Ionic Strength:
Conclusion
Understanding chemical equilibrium, solubility, acid-base chemistry, and the role of activities and ionic strength is fundamental for analytical chemistry. Mastery of these concepts enables accurate prediction and control of chemical systems in both laboratory and real-world applications.
