 Back
BackElectrochemistry, Redox, and Analytical Measurements Exam Guidance
Study Guide - Smart Notes

Q1. Assign Oxidation States to Underlined Elements in Compounds
Background
Topic: Oxidation States and Redox Chemistry
This question tests your ability to assign oxidation numbers to specific elements in various compounds, a foundational skill for understanding redox reactions and balancing equations in analytical chemistry.
Key Terms and Concepts:
Oxidation State: The hypothetical charge an atom would have if all bonds to atoms of different elements were 100% ionic.
Rules for Assigning Oxidation States:
Elements in their standard state: 0
Group 1 metals: +1; Group 2 metals: +2
Oxygen: usually -2 (except in peroxides, where it is -1)
Hydrogen: +1 (with nonmetals), -1 (with metals)
Sum of oxidation states in a neutral compound: 0; in a polyatomic ion: equals the ion charge
Step-by-Step Guidance
For each compound, write the chemical formula and identify the element whose oxidation state you need to determine.
Apply the rules above to assign oxidation states to the other elements in the compound.
Set up an equation where the sum of all oxidation states equals the overall charge of the molecule or ion.
Solve for the unknown oxidation state.
Try solving on your own before revealing the answer!
Q2. Electrochemical Cell Line Notation and Calculations
Background
Topic: Electrochemical Cells, Standard Potentials, and Thermodynamics
This question assesses your understanding of cell notation, writing half-reactions, calculating cell potentials, Gibbs free energy, and equilibrium constants for redox reactions.
Key Terms and Formulas:
Cell Notation: Anode | Anode solution || Cathode solution | Cathode
Standard Cell Potential:
Gibbs Free Energy:
Equilibrium Constant:
Step-by-Step Guidance
Identify the half-reactions at each electrode using the cell notation and standard reduction potentials.
Write the reduction half-reactions for both the anode and cathode.
Combine the half-reactions to write the balanced net cell reaction.
Use the standard reduction potentials to calculate .
Set up the equations for and the equilibrium constant using the relationships above.
Try solving on your own before revealing the answer!
Q3. Determining the Acid Dissociation Constant () from Cell Voltage
Background
Topic: Electrochemical Determination of Acid Strength
This question tests your ability to relate cell voltage to equilibrium concentrations and calculate the acid dissociation constant for a weak acid using the Nernst equation.
Key Terms and Formulas:
Nernst Equation:
Reaction Quotient (): Ratio of product and reactant concentrations raised to their stoichiometric coefficients.
Acid Dissociation Constant ():
Step-by-Step Guidance
Write the half-cell reactions and identify the overall cell reaction.
Calculate the standard cell potential using the given values.
Set up the Nernst equation for the cell, substituting the given concentrations and voltage.
Express the reaction quotient in terms of , , and .
Rearrange the equation to solve for in terms of the measured cell voltage and concentrations.
Try solving on your own before revealing the answer!
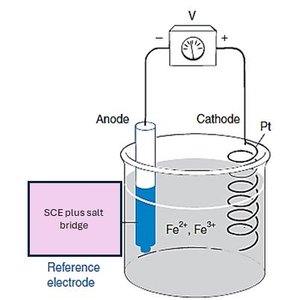
Q4. Voltaic Cell with SCE Reference Electrode
Background
Topic: Reference Electrodes and Cell Potentials
This question evaluates your understanding of how to use a Saturated Calomel Electrode (SCE) as a reference and calculate the cell voltage based on ion concentrations and standard potentials.
Key Terms and Formulas:
SCE (Saturated Calomel Electrode): A common reference electrode with a known potential relative to the Standard Hydrogen Electrode (SHE).
Nernst Equation:
Cell Voltage:
Step-by-Step Guidance
Identify the half-cell reactions and assign the SCE as the reference (anode or cathode as appropriate).
Write the Nernst equation for the Fe3+/Fe2+ couple, using the given concentrations.
Calculate the electrode potential for the Fe3+/Fe2+ half-cell.
Determine the cell voltage by referencing the SCE potential (given as 0.241 V vs. SHE).
Try solving on your own before revealing the answer!
Q5. pH Electrode Calibration and Sample Measurement
Background
Topic: pH Measurement and Electrode Calibration
This question tests your ability to calibrate a pH electrode using standard buffers, determine the slope and correction factor, and calculate the pH of an unknown solution from a voltage reading.
Key Terms and Formulas:
Calibration Slope:
Theoretical Slope:
Correction Factor ():
pH Calculation:
Step-by-Step Guidance
Calculate the observed slope using the voltage and pH values of the two standard buffers.
Determine the correction factor by dividing the observed slope by the theoretical slope.
Set up the calibration equation relating voltage to pH using the slope and one of the calibration points.
Rearrange the equation to solve for the pH of the unknown sample given its voltage reading.
Try solving on your own before revealing the answer!
Final Answers
Q1: (1) Mn: +7 in KMnO4, (2) Mn: +4 in MnO2, (3) Cr: +6 in K2Cr2O7, (4) Al: +3 in Al(OH)3, (5) O: -1 in H2O2
Q2: (1) Fe3+ + e- → Fe2+ (cathode), MnO4- + 8H+ + 5e- → Mn2+ + 4H2O (anode); (2) Fe2+ + MnO4- + H+ → Fe3+ + Mn2+ + H2O (balanced); (3) E°cell = 0.77 - 1.51 = -0.74 V; (4) ΔG° = -nFE°cell; (5) K = exp(nFE°cell/RT)
Q3: KA = 0.005 × [H+]/0.050, with [H+] found from Nernst equation using cell voltage and concentrations.
Q4: (1) E(cathode) = 0.771 V - (0.05916/1) × log([Fe2+]/[Fe3+]); (2) Ecell = E(cathode) - 0.241 V
Q5: (1) Slope = (146.3 - (-18.3))/(4.002 - 6.881) mV/pH; (2) β = observed slope/59.16; (3) Use calibration equation to solve for pH when E = +100.0 mV

