 Back
BackExtraction Techniques in Analytical Chemistry: Solid-Liquid, Liquid-Liquid, and Solid Phase Extraction
Study Guide - Smart Notes

Extraction Techniques in Analytical Chemistry
Introduction to Extraction Methods
Extraction techniques are fundamental in analytical chemistry for isolating analytes from complex matrices, such as pharmaceutical formulations, biological samples, and natural products. These methods enhance the accuracy and reliability of subsequent analyses by removing interfering substances.
Extraction refers to the process of separating a substance from a mixture using a suitable solvent.
Common contexts include product purification, drug extraction from natural sources, and sample preparation for analysis.
Major extraction types: solid-liquid extraction, liquid-liquid extraction, and solid phase extraction (SPE).
Solid-Liquid Extraction
Principles and Applications
Solid-liquid extraction involves transferring analytes from a solid matrix into a liquid solvent. This technique is widely used for isolating active compounds from natural sources and purifying products after synthesis.
Key applications: Extraction of drugs from plants (e.g., salicylin from willow, morphine from opium poppy, digoxin from foxglove, paclitaxel from yew, artemisinin from sweet wormwood).
Process: The solid sample is mixed with a solvent in which the analyte is highly soluble, while matrix components are less soluble.
Further solvent partitioning may be required to reduce interferents.
Example: Extraction of salicylin from Salix alba (white willow) as a precursor to aspirin.

Example: Extraction of morphine from Papaver somniferum (opium poppy).

Example: Extraction of digoxin from Digitalis lanata (foxglove).

Example: Extraction of paclitaxel from Taxus brevifolia (Pacific yew).

Example: Extraction of artemisinin from Artemisia annua (sweet wormwood).

Continuous Extraction: Soxhlet Extraction
Continuous extraction methods, such as the Soxhlet extractor, allow for efficient extraction of analytes from solids using repeated washing with fresh solvent.
Soxhlet extraction: The sample is placed in a thimble and repeatedly washed with boiling solvent, which condenses and percolates through the sample.
Ensures exhaustive extraction without the need for large solvent volumes.
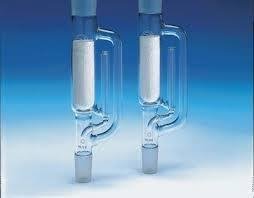
Liquid-Liquid Extraction (LLE)
Principles and Applications
Liquid-liquid extraction separates analytes based on their differential solubility in two immiscible liquids, typically an aqueous and an organic phase. It is commonly used for sample clean-up before chromatographic analysis.
Partition coefficient (P): Quantifies the distribution of an analyte between two phases.
Simple equipment: separatory funnel, solvents, and collection vessels.
Steps: Mix sample with extraction solvent, allow phases to separate, collect desired layer, and evaporate solvent if needed.
Example: Extraction of organic acids and bases. Free acids/bases are soluble in organic solvents, while their salts are water-soluble.
~20% of drugs are weak acids (often as sodium/potassium salts).
~75% of drugs are weak bases (often as hydrochloride salts).
Flow Diagram for Extraction of an Organic Base
This diagram illustrates the sequential steps for extracting organic bases from complex mixtures, including acidification, basification, and selective solvent extraction.
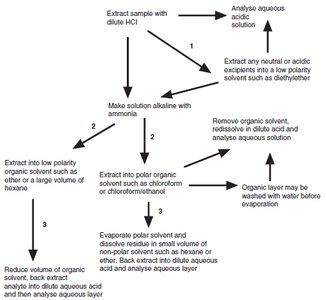
Partitioning Between Organic Solvents
Partitioning between organic solvents is used for analytes in oily excipients, such as steroid creams. Specific solvent mixtures (e.g., methanol/hexane) are chosen based on analyte and matrix properties.
Pharmacopoeia assays for corticosteroids use such methods.
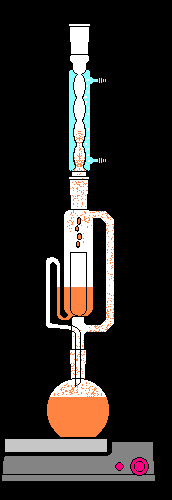
Continuous Liquid-Liquid Extraction
Continuous liquid-liquid extraction setups allow for the efficient extraction of analytes when the partition coefficient is low or when large sample volumes are involved.
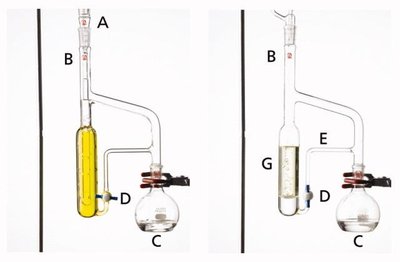
Supercritical Fluid Extraction (SFE)
Principles and Advantages
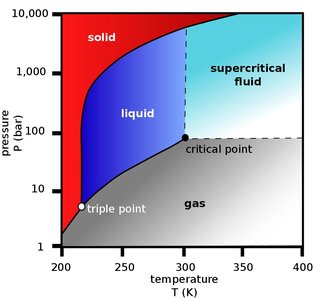
Supercritical fluid extraction uses fluids above their critical temperature and pressure, where they exhibit both gas-like and liquid-like properties. The most common supercritical fluid is carbon dioxide (CO2).
Advantages: Faster mass transfer, tunable solvent power (by adjusting pressure), and the ability to add polar modifiers (e.g., methanol, formic acid).
CO2 is non-toxic, non-flammable, and has a low critical temperature (31.1°C at 73.8 bar).
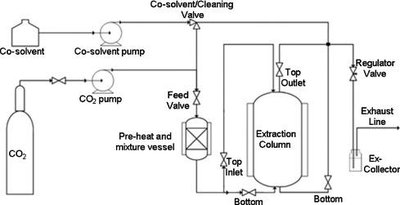
Schematic of SFE Equipment
The schematic shows the main components of an SFE system, including the CO2 source, pumps, extraction column, and collection system.
Solid Phase Extraction (SPE)
Principles and Steps
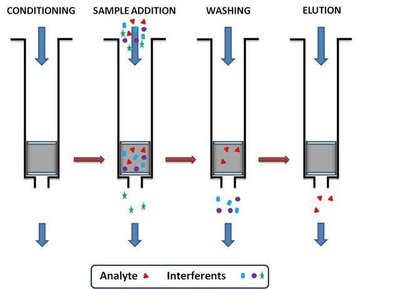
Solid phase extraction is a sample preparation technique where analytes are retained on a solid adsorbent and selectively eluted. SPE is widely used for cleaning up biological and environmental samples.
Steps:
Conditioning the SPE medium
Sample loading (analyte is retained)
Washing (removes contaminants)
Elution (analyte is recovered in a small solvent volume)
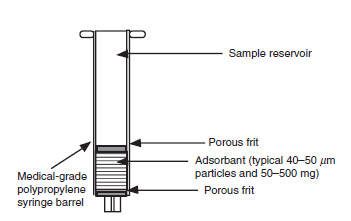
SPE Cartridge Design
The SPE cartridge is the core component, containing the adsorbent material and frits to retain particles.
Real-World SPE Examples
SPE is implemented using cartridges and manifolds for batch processing and automation.


Types of SPE Adsorbents
The choice of adsorbent determines the selectivity and efficiency of SPE. Main types include:
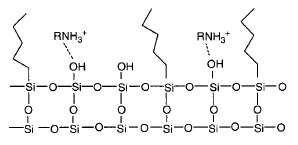
Lipophilic surfaces: Coated silica gels, styrene-divinylbenzene polymers (for non-polar analytes).
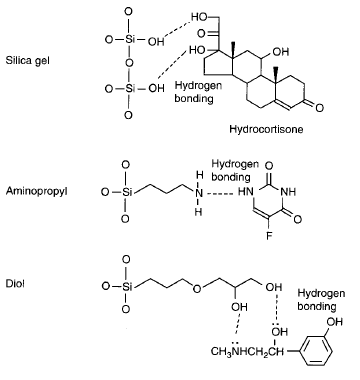
Polar surfaces: Polar surface-modified silica gels (for polar compounds).
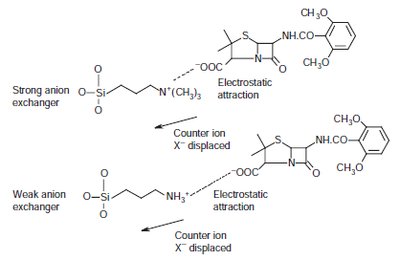
Ion exchangers: Modified silica gels for anion or cation exchange.
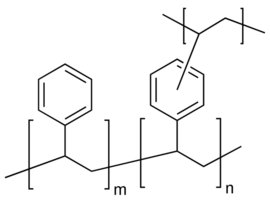
Selective materials: Borate gels (for diols), immunoaffinity gels (antibody-based), and molecularly imprinted polymers (MIPs).

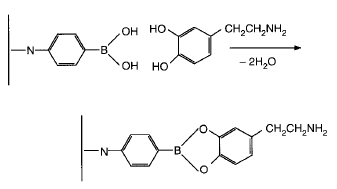
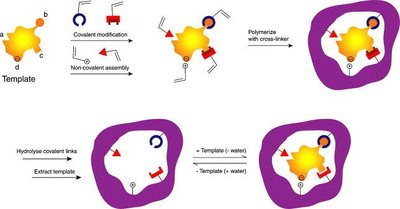
Applications of Molecularly Imprinted Polymers (MIPs)
MIPs are synthetic polymers with selective binding sites for target analytes, acting as 'plastic antibodies.' They are commercially available for a range of substances, including toxins, drugs, and environmental contaminants.

Strengths and Limitations of SPE
Strengths | Limitations |
|---|---|
Solid phase is immiscible with solvents | Silica gel-based adsorbents are unstable to strong acids/bases |
Variety of adsorbent chemistries | Cartridge cost |
No emulsion formation | Use of internal standards is advisable |
Sample concentration possible | — |
Low solvent consumption | — |
Batch processing and automation | — |
Summary and Take-Home Points
Extraction techniques are essential for isolating analytes from complex matrices in analytical chemistry.
Solid-liquid and liquid-liquid extraction are foundational methods, with SPE offering enhanced selectivity and automation.
The choice of adsorbent and extraction method depends on analyte properties and sample matrix.
Modern materials, such as MIPs, expand the selectivity and application range of SPE.
