 Back
BackInfrared Spectroscopy in Analytical Chemistry: Principles, Interpretation, and Applications
Study Guide - Smart Notes

Infrared Spectroscopy: Principles and Applications
Introduction to Infrared (IR) Spectroscopy
Infrared spectroscopy is a fundamental analytical technique used to study the interaction of electromagnetic radiation with matter, specifically in the infrared region. It is widely applied in pharmaceutical analysis for structural confirmation and identification of compounds.
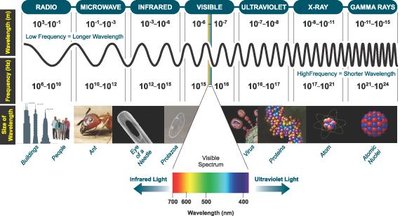
Electromagnetic Spectrum: IR spectroscopy focuses on the region between 800 nm and 1 nm, with the mid-infrared (4,000–625 cm-1) most commonly used for structural analysis.
Applications: Used for qualitative fingerprinting, compound identification, and characterization of solids, semi-solids, and polymorphs in pharmaceuticals.
Principles of IR Spectroscopy
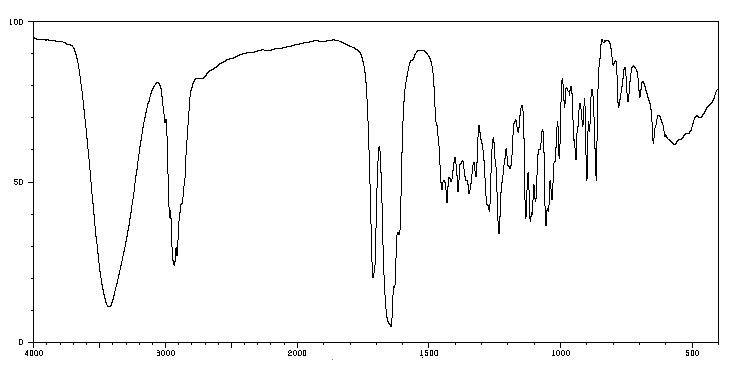
IR spectroscopy measures the absorption of IR radiation by molecules, which causes vibrational transitions in chemical bonds. Each bond and functional group absorbs at characteristic frequencies, producing a unique spectrum.
IR-active Bonds: Only bonds between atoms with different electronegativities (partial charges) absorb IR radiation. For example, N2 and O2 do not show IR spectra.
Dipole Moment: The intensity of absorption depends on the dipole moment, which is influenced by the relative electronegativities of the atoms.
Vibrational Modes: Molecules exhibit stretching (symmetrical and asymmetrical) and bending (rocking, scissoring, twisting, wagging, out-of-plane) vibrations.
Hooke’s Law and Vibrational Frequencies
The frequency of vibration for a chemical bond can be approximated using Hooke’s Law, which models the bond as a spring:
Equation:
Parameters: k = force constant (bond strength), m = reduced mass ()
Bond Strength: Double and triple bonds have higher force constants and vibrate at higher frequencies than single bonds.
Reduced Mass: Lighter atoms (e.g., O-H) vibrate at higher frequencies than heavier atoms (e.g., C-O).


Characteristic IR Absorptions of Functional Groups
Functional groups absorb IR radiation at specific, predictable regions, allowing for identification and structural analysis.
Single Bonds to Hydrogen: The position is determined by bond strength and hybridization state.
Triple Bonds: C≡C and C≡N stretches appear in the 2100–2200 cm-1 region.
Double Bonds: C=C and C=O stretches are found in the 1600–1800 cm-1 region.
Carbonyl Region: The C=O stretch varies depending on the compound type (e.g., ketone, aldehyde, ester, acid).
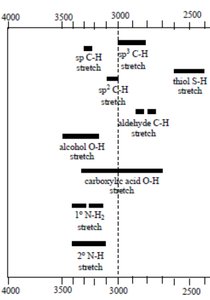
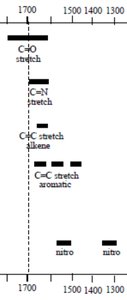

Interpretation of IR Spectra: Examples
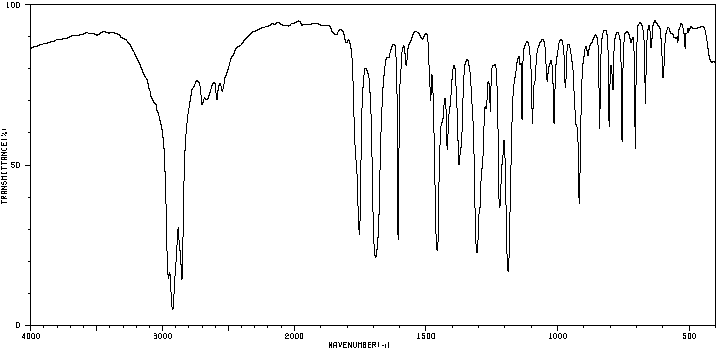
IR spectra provide a fingerprint for each compound, with characteristic peaks corresponding to functional groups.
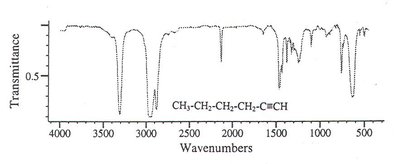
Alkane: Only C-H and C-C bonds, weak absorptions.
Alkene: Additional bands for =C-H and C=C stretches.
Alkyne: C≡C stretch (2100–2200 cm-1), ≡C-H stretch (~3300 cm-1).
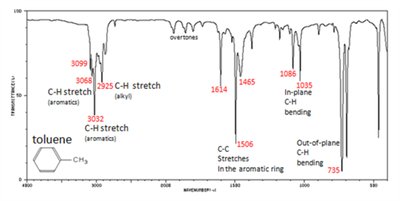
Aromatic Compound: Complex pattern due to ring vibrations.
Ketone: Strong C=O absorption near 1700 cm-1.
Aldehyde: C=O absorption at a different position, additional C-H stretches.
Alcohol: Broad O-H absorption near 3300 cm-1.
Aspirin: Multiple characteristic peaks for carboxylic OH, C-H, C=O (ester and acid), and aromatic C=C stretches.
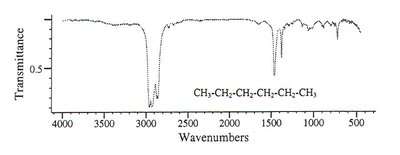
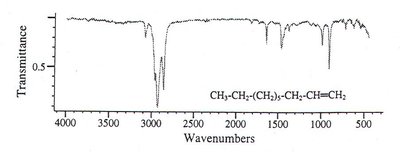
Applications of IR Spectroscopy in Pharmaceutical Analysis
IR spectroscopy is a versatile tool in pharmaceutical analysis, providing rapid and reliable identification of compounds and materials.
Qualitative Fingerprint: Used to verify the identity of raw materials and finished products.
Synthetic Chemistry: Preliminary check for compound identity and functional group presence.
Characterization: Used for solids, semi-solids, creams, and tablets.
Polymorph Detection: Identifies different crystal forms (polymorphs) with distinct physical properties.
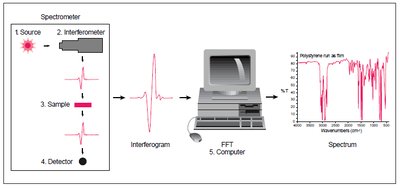
Instrumentation for IR Spectroscopy
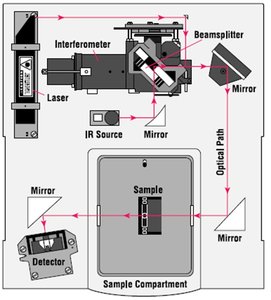
Two main types of IR spectrometers are used: dispersive (monochromator-based) and Fourier Transform (interferometer-based). FT-IR instruments are faster and more widely used.
Dispersive Instruments: Use a monochromator to separate IR wavelengths.
Fourier Transform Instruments: Use an interferometer and computer to rapidly acquire spectra.
Sample Preparation Methods
Proper sample preparation is essential for accurate IR measurements. Methods vary depending on the sample type.
Thin Film: Created between two NaCl discs for liquids or solids (paraffin as solvent).
KBr Disc: Used for solid samples.
Cells: Used for liquids or solutions.
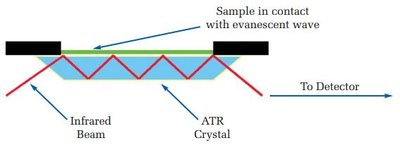
Attenuated Total Reflectance (ATR): Allows direct analysis of solids, liquids, and semi-solids.


Advantages and Limitations of IR Spectroscopy
IR spectroscopy offers unique advantages but also has limitations in analytical chemistry.
Strengths: Provides a unique fingerprint for each molecule; spectra can be matched to standards using computers.
Limitations: Rarely used quantitatively due to complex spectra; only detects gross impurities; sample preparation can be challenging.
Summary of Learning Outcomes
General principles of IR spectroscopy
Characteristics and interpretation of IR spectra
Absorption regions for different functional groups
Instrumentation and sample preparation methods
Applications in pharmaceutical analysis
