 Back
BackAcid-Base Balance: Bicarbonate Buffer System and Acid-Base Disturbances
Study Guide - Smart Notes

Q10. How does the equation below shift when the plasma CO2 concentration rises?
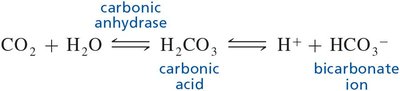
Background
Topic: Acid-Base Balance & Buffer Systems
This question tests your understanding of the bicarbonate buffer system, which is crucial for maintaining acid-base balance in the body. The equation shows how carbon dioxide (CO2) is converted to carbonic acid (H2CO3), which then dissociates into hydrogen ions (H+) and bicarbonate ions (HCO3-).
Key Terms and Formulas
Bicarbonate Buffer System Equation:
Le Chatelier's Principle: If the concentration of a reactant increases, the equilibrium shifts to reduce that change (i.e., toward the products).
Acid-Base Disturbances: Changes in CO2 or HCO3- can lead to respiratory or metabolic acidosis/alkalosis.
Step-by-Step Guidance
Examine the equation:
Recall that an increase in CO2 means more reactant on the left side of the equation.
Apply Le Chatelier's Principle: The system will shift to the right to use up the excess CO2, producing more H2CO3, H+, and HCO3-.
Consider the physiological effect: Increasing H+ will lower pH, leading to acidosis if not compensated.
Try solving on your own before revealing the answer!
Final Answer: The equation shifts to the right.
When plasma CO2 rises, the equilibrium shifts to produce more H2CO3, which then dissociates into H+ and HCO3-. This can result in a decrease in blood pH (acidosis) if not compensated by the respiratory or renal systems.
