 Back
BackAcid-Base Balance in Human Physiology
Study Guide - Smart Notes

Acid-Base Balance
Overview of Acid-Base Balance
The maintenance of acid-base balance is essential for normal cellular function and overall homeostasis. The body tightly regulates blood pH within a narrow range to ensure optimal enzyme activity and physiological processes.
Normal blood pH: 7.35–7.45
Arterial blood pH: 7.4
Venous blood pH: 7.35
Alkalosis: Arterial blood pH > 7.45 (results in alkalemia)
Acidosis: Arterial blood pH < 7.35 (results in acidemia)
Hydrogen Ion Regulation
Mechanisms of Regulation
The concentration of hydrogen ions (H+) in the body is regulated by three main systems, each acting at different speeds:
Chemical buffer systems: Act within seconds to resist changes in pH.
Bicarbonate buffer system: H2CO3/HCO3-
Protein buffers: e.g., hemoglobin
Respiratory system: Acts within 1–3 minutes by adjusting CO2 elimination.
Renal (urinary) system: Requires hours to days to change pH by excreting or reabsorbing acids and bases.
Acid-Base Regulation by the Respiratory System
Role of CO2 in pH Regulation
The respiratory system helps regulate blood pH by controlling the amount of carbon dioxide (CO2) exhaled. CO2 combines with water to form carbonic acid, which dissociates into hydrogen ions and bicarbonate:
Acidosis compensation: Increased respiratory rate and depth eliminate more CO2, lowering H+ and raising blood pH.
Alkalosis compensation: Decreased respiratory rate and depth retain CO2, increasing H+ and lowering blood pH.
Acid-Base Regulation by the Renal System
Renal Compensation Mechanisms
The kidneys are the ultimate regulators of acid-base balance, capable of excreting or reabsorbing acids and bases as needed:
Acidosis compensation: Kidneys reabsorb more HCO3- and secrete more H+ to increase pH.
Alkalosis compensation: Kidneys reabsorb more H+ and secrete more HCO3- to decrease pH.
Chemical buffers and the lungs can only temporarily tie up or eliminate acids and bases, but only the kidneys can remove metabolic acids (e.g., phosphoric, uric, lactic acids, and ketones) from the body.
Types of Acid-Base Imbalances
Respiratory Acidosis
Definition: Blood pH < 7.35 due to decreased CO2 elimination (hypoventilation).
Causes: Shallow breathing, impaired gas exchange (pneumonia, cystic fibrosis, emphysema), or respiratory depression (narcotic/barbiturate overdose).
Respiratory Alkalosis
Definition: Blood pH > 7.45 due to increased CO2 elimination (hyperventilation).
Causes: Rapid or shallow breathing from pain, anxiety, or asthma.
Metabolic Acidosis
Definition: Blood pH < 7.35 due to increased acid accumulation or bicarbonate loss.
Causes of acid accumulation: Excess alcohol, aspirin, exercise, untreated diabetes mellitus, starvation, renal disease.
Causes of bicarbonate loss: Excessive diarrhea, vomiting from lower GI tract.
Metabolic Alkalosis
Definition: Blood pH > 7.45 due to increased bicarbonate reabsorption or hydrogen ion loss.
Causes of bicarbonate reabsorption: Excessive antacid ingestion, constipation.
Causes of hydrogen ion loss: Vomiting from upper GI tract, diuretic use (leading to aldosterone secretion).
Physiological Effects of Acidosis and Alkalosis
Impact on the Central Nervous System (CNS)
Acidosis: CNS depression (muscle and nerve cells are hyperpolarized), leading to confusion, coma, and potentially death.
Alkalosis: CNS overstimulation (muscle and nerve cells are easily depolarized due to excess K+), resulting in muscle tetany, nervousness, convulsions, respiratory paralysis, and potentially death.
Potassium Balance and Acid-Base Disorders
Relationship Between Acid-Base Status and Potassium Levels
Acid-base imbalances can significantly affect potassium (K+) distribution between the intracellular and extracellular compartments:
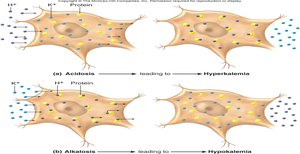
Acidosis: H+ ions move into cells, causing K+ to move out, leading to hyperkalemia (elevated blood potassium).
Alkalosis: H+ ions move out of cells, causing K+ to move in, leading to hypokalemia (reduced blood potassium).
Summary Table: Acid-Base Disorders
Disorder | Blood pH | Primary Cause | Compensation |
|---|---|---|---|
Respiratory Acidosis | < 7.35 | CO2 retention (hypoventilation) | Renal: ↑ HCO3- reabsorption, ↑ H+ secretion |
Respiratory Alkalosis | > 7.45 | CO2 loss (hyperventilation) | Renal: ↑ H+ reabsorption, ↑ HCO3- secretion |
Metabolic Acidosis | < 7.35 | Acid accumulation or HCO3- loss | Respiratory: ↑ ventilation; Renal: ↑ HCO3- reabsorption |
Metabolic Alkalosis | > 7.45 | HCO3- reabsorption or H+ loss | Respiratory: ↓ ventilation; Renal: ↑ H+ reabsorption |
Example: A patient with severe diarrhea (loss of HCO3-) may develop metabolic acidosis, while a patient with prolonged vomiting (loss of H+ from the stomach) may develop metabolic alkalosis.
Additional info: The interplay between acid-base balance and electrolyte (especially potassium) homeostasis is clinically significant, as disturbances can lead to life-threatening cardiac and neuromuscular complications.
