 Back
BackAcid–Base Balance and pH Regulation in the Human Body
Study Guide - Smart Notes

Hydrogen Ions (H+) and pH
Definition and Importance of H+ in Biological Systems
The concentration of hydrogen ions (H+) in aqueous solutions is a fundamental aspect of cellular and systemic physiology. All water-based solutions, including those in the human body (intracellular fluid, plasma, interstitial fluid), contain some H+ ions. The regulation of H+ is critical because these ions are highly reactive and can disrupt the structure and function of proteins and other biomolecules.
Acid: A substance that increases the concentration of H+ ions when added to a solution.
Base (Alkali): A substance that decreases the concentration of H+ ions when added to a solution.
Even pure water contains H+ due to the dissociation reaction:
In pure water, [H+] ≈ M.
Excess H+ can denature proteins, especially enzymes and channel proteins, making them non-functional.
pH: Measurement of Hydrogen Ion Concentration
pH is the standard method for expressing the concentration of H+ in a solution. It is defined as the negative logarithm (base 10) of the H+ concentration:
Lower pH values indicate higher H+ concentrations (more acidic).
Higher pH values indicate lower H+ concentrations (more basic or alkaline).
Examples:
pH 2 = 0.01 M [H+]
pH 5 = 0.00001 M [H+]
pH 10 = 0.0000000001 M [H+]
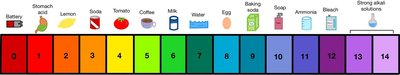
Acid–Base Balance in the Human Body
pH Scale and Physiological Ranges
The pH scale ranges from 0 (most acidic) to 14 (most basic). In the human body, the homeostatic value for plasma pH is approximately 7.4 (typically 7.38–7.42). Deviations from this range can have significant physiological consequences.
Acidosis: Plasma pH < 7.35 (excess H+); can cause decreased CNS activity, muscle weakness, arrhythmias, and coma.
Alkalosis: Plasma pH > 7.45 (deficient H+); can cause increased CNS activity, tingling, muscle cramps, and respiratory paralysis.
Neutral: pH = 7 (equal [H+] as pure water).

Sources of Acids and Bases in the Body
Acids and bases in the body originate from various sources:
Dietary acids: Amino acids, fatty acids, etc.
Metabolic acids: Lactic acid (anaerobic metabolism), ketoacids (lipid breakdown).
CO2: The largest source; CO2 dissolves in blood and forms H+ and HCO3- ().
Bases are rare in diet/metabolism; the body primarily eliminates excess acids.
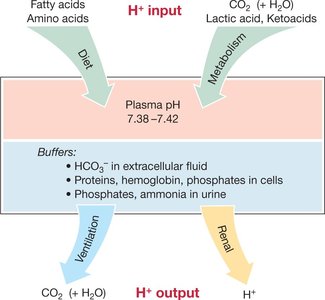
Mechanisms of pH Homeostasis
Buffers, Lungs, and Kidneys
The body maintains pH homeostasis through three main mechanisms:
Buffers: Substances that minimize pH changes by absorbing or releasing H+ as needed.
Bicarbonate (HCO3-): The most important buffer in plasma. Reaction:
Other buffers: Proteins, hemoglobin, phosphates, ammonia.
Buffers do not eliminate H+, but reduce the impact of acid/base addition.
Lungs (Ventilation): Regulate CO2 levels, thus affecting H+ concentration.
Increased ventilation (hyperventilation): Decreases CO2, decreases H+, raises pH.
Decreased ventilation (hypoventilation): Increases CO2, increases H+, lowers pH.
Allows rapid response to pH changes, but limited by respiratory needs.
Kidneys: Regulate excretion of H+ and HCO3- in urine.
Low pH: Kidneys excrete H+, reabsorb HCO3-.
High pH: Kidneys retain H+, excrete HCO3-.
Slower but essential for long-term acid-base balance.
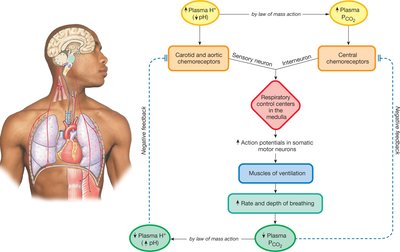
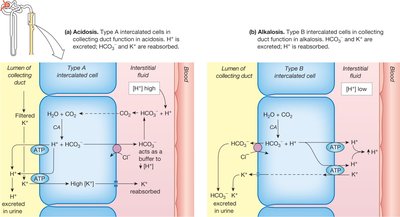
Acid–Base Disturbances: Classification and Diagnosis
Types of Acid–Base Disturbances
Acid–base disturbances are classified by their origin:
Respiratory disturbances: Caused by changes in CO2 levels.
Respiratory acidosis: Too much CO2 (e.g., emphysema, hypoventilation).
Respiratory alkalosis: Too little CO2 (e.g., hyperventilation).
Metabolic disturbances: Caused by non-respiratory factors.
Metabolic acidosis: Excess acid from metabolism (e.g., diabetic ketoacidosis).
Metabolic alkalosis: Excess base or loss of acid (e.g., vomiting, loss of gastric acid).
Diagnosis Using Blood Values
Measuring the partial pressure of CO2 (PCO2) and the concentration of HCO3- in the blood helps distinguish between respiratory and metabolic causes:
Disturbance | pH | CO2 | HCO3- |
|---|---|---|---|
Respiratory Acidosis | Low | High | High |
Metabolic Acidosis | Low | Low | Low |
Respiratory Alkalosis | High | Low | Low |
Metabolic Alkalosis | High | High | High |
Summary Table: Plasma PCO2, Ions, and pH in Acid–Base Disturbances
The following table summarizes the typical changes in plasma PCO2, HCO3-, and pH during different acid–base disturbances:
Condition | pH | PCO2 | HCO3- |
|---|---|---|---|
Respiratory Acidosis | Low | High | High |
Metabolic Acidosis | Low | Low | Low |
Respiratory Alkalosis | High | Low | Low |
Metabolic Alkalosis | High | High | High |
Key Equations and Concepts
pH calculation:
Water dissociation:
Bicarbonate buffer system:
Additional info: The above notes integrate textbook-level explanations, relevant equations, and visual aids to provide a comprehensive overview of acid–base balance and regulation in the human body, suitable for ANP college students.
