 Back
BackAdrenal Cortex: Structure, Function, and Clinical Relevance
Study Guide - Smart Notes

Adrenal Cortex: Structure and Function
Overview of the Adrenal Gland
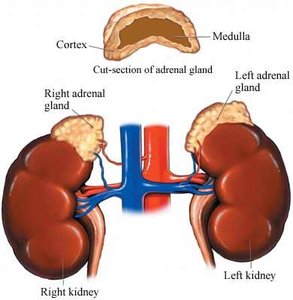
The adrenal glands are paired endocrine organs located on top of the kidneys in mammals. Each gland consists of two distinct regions: the outer adrenal cortex and the inner adrenal medulla. These regions are functionally and structurally different, with the cortex primarily responsible for steroid hormone production.
Adrenal cortex: Produces steroid hormones (glucocorticoids, mineralocorticoids, and androgens).
Adrenal medulla: Produces catecholamines (epinephrine and norepinephrine).
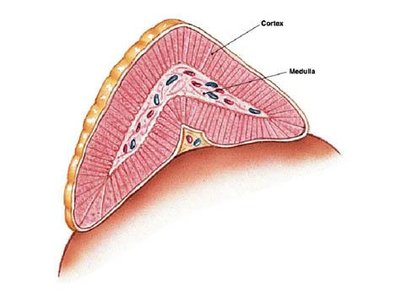
Histological Structure of the Adrenal Cortex
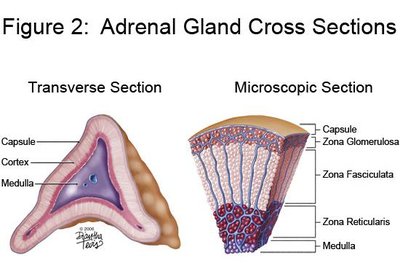
The adrenal cortex is divided into three concentric zones, each responsible for the synthesis of different steroid hormones. From the outermost to innermost:
Zona glomerulosa: Produces mineralocorticoids (mainly aldosterone).
Zona fasciculata: Produces glucocorticoids (mainly cortisol).
Zona reticularis: Produces androgens (mainly DHEA and DHEA-S).
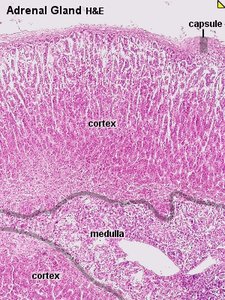
Adrenal Cortex Steroidogenesis
Steroid Hormone Classes and Functions
The adrenal cortex synthesizes three main classes of steroid hormones, all derived from cholesterol:
Glucocorticoids (C21): Regulate metabolism and immune response (e.g., cortisol).
Mineralocorticoids (C21): Regulate sodium and potassium balance (e.g., aldosterone).
Androgens (C19): Weak sex steroids (e.g., DHEA, DHEA-S).
Steroidogenic cells contain abundant lipid droplets (for cholesterol storage), mitochondria, and smooth endoplasmic reticulum (SER), which are essential for steroid biosynthesis.
Cholesterol as the Precursor
Cholesterol (C27) is the substrate for all steroid hormones. It is transported into mitochondria, where the terminal six carbons are cleaved to form pregnenolone (C21), the first steroid in the pathway.
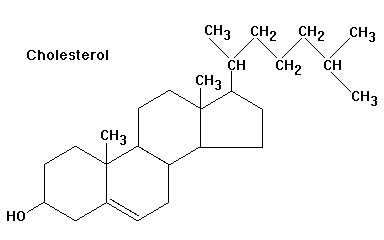
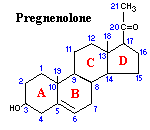
Pathways and Enzymes of Steroid Biosynthesis
The conversion of cholesterol to various steroid hormones involves several key enzymes, each localized to specific zones of the cortex. The main steps include:
Side-chain cleavage (CYP11A1/desmolase): Cholesterol → Pregnenolone
3β-hydroxysteroid dehydrogenase (3βHSD): Pregnenolone → Progesterone
17α-hydroxylase/17,20-lyase (CYP17): For glucocorticoid and androgen synthesis
21-hydroxylase (CYP21A2) and 11β-hydroxylase (CYP11B1): For glucocorticoid and mineralocorticoid synthesis
Aldosterone synthase (CYP11B2): For aldosterone synthesis (zona glomerulosa only)
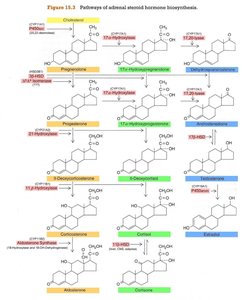
Zone-Specific Steroid Production
Each zone of the adrenal cortex expresses a unique set of enzymes, determining which hormones are produced:
Zona glomerulosa: Lacks CYP17, produces only mineralocorticoids (aldosterone).
Zona fasciculata and reticularis: Produce glucocorticoids and androgens, but not aldosterone (lack aldosterone synthase).
Regulation of Adrenal Cortex Function
Regulation of Aldosterone (Zona Glomerulosa)
Aldosterone secretion is primarily regulated by:
Renin-angiotensin system (Angiotensin II): Stimulates aldosterone release in response to low blood pressure or low sodium.
Plasma potassium (K+): High K+ directly stimulates aldosterone secretion.
Regulation of Cortisol and Androgens (Zona Fasciculata and Reticularis)
Glucocorticoid and androgen secretion is regulated by the hypothalamic-pituitary-adrenal (HPA) axis:
CRH (Corticotropin-releasing hormone): Released from the hypothalamus in response to stress or circadian signals.
ACTH (Adrenocorticotropic hormone): Released from the anterior pituitary, stimulates the adrenal cortex.
Negative feedback: Cortisol inhibits CRH and ACTH release.
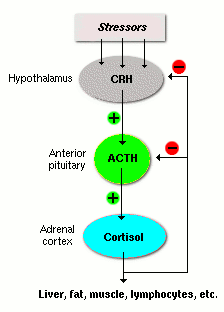
Circadian and Stress Regulation
ACTH and cortisol are secreted in a circadian rhythm, peaking in the early morning. Stress can trigger rapid increases in ACTH and cortisol.
Physiological Actions of Adrenal Cortex Hormones
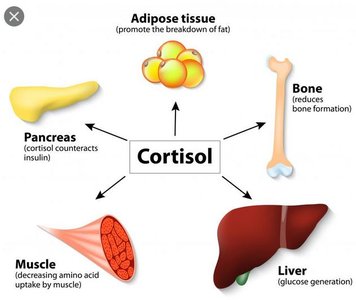
Glucocorticoids (Cortisol)
Cortisol is the primary glucocorticoid in humans, affecting nearly all tissues:
Metabolic effects: Increases gluconeogenesis in the liver, mobilizes amino acids from muscle, and promotes lipolysis in adipose tissue.
Catabolic effects: Inhibits glucose uptake in muscle and adipose tissue, leading to protein and fat breakdown.
Permissive effects: Enhances the actions of catecholamines and other hormones during stress.
Anti-inflammatory and immunosuppressive effects: Suppresses immune cell activity and inflammation.
Mineralocorticoids (Aldosterone)
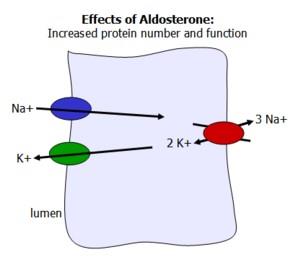
Aldosterone acts on the distal tubules of the kidney to increase sodium reabsorption and potassium excretion, thereby regulating blood volume and pressure.
Increases expression and activity of Na+/K+ ATPase and epithelial sodium channels (ENaC).
Promotes water retention via osmotic effects of sodium.
Androgens (DHEA, DHEA-S)
Adrenal androgens are weak sex steroids, with DHEA-S being the major circulating form. They contribute to the development of secondary sexual characteristics, especially in females and during adrenarche.
Development of the Adrenal Cortex
Fetal and Postnatal Development
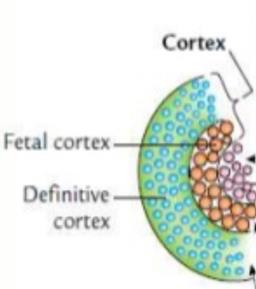
The adrenal cortex develops from mesodermal tissue in the genital ridge. In the fetus, the gland is much larger relative to body weight and consists of a large fetal zone and a smaller permanent zone. After birth, the fetal zone regresses, and the adult zones differentiate over the first few years of life.
Pathophysiology of Adrenal Cortex Disorders
Adrenocortical Hypofunction (Addison's Disease)
Primary adrenal insufficiency (Addison's disease) results from destruction of the adrenal cortex, leading to deficiencies in glucocorticoids, mineralocorticoids, and androgens. Symptoms include:
Weakness, weight loss, hypotension
Hyperpigmentation (due to increased ACTH)
Salt craving, hypoglycemia
Adrenocortical Hyperfunction (Cushing's Syndrome and Conn's Syndrome)
Excessive secretion of adrenal cortex hormones can result from tumors or overproduction of ACTH:
Cushing's syndrome: Excess cortisol; symptoms include centripetal obesity, muscle weakness, hypertension, and skin changes.
Conn's syndrome: Excess aldosterone; symptoms include hypertension, hypokalemia, and metabolic alkalosis.

Summary Table: Zones, Hormones, and Functions
Zone | Hormone(s) | Main Function | Example(s) |
|---|---|---|---|
Zona glomerulosa | Mineralocorticoids | Regulate mineral balance | Aldosterone |
Zona fasciculata | Glucocorticoids | Regulate glucose metabolism | Cortisol |
Zona reticularis | Androgens | Stimulate masculinization | DHEA, DHEA-S |
