 Back
BackDrug Metabolism: Phase I Reactions and Cytochrome P450 Enzymes
Study Guide - Smart Notes

Drug Metabolism
Introduction to Drug Metabolism
Drug metabolism refers to the biochemical modification of pharmaceutical substances by living organisms, usually through specialized enzymatic systems. The process is essential for the detoxification and elimination of drugs from the body, primarily occurring in the liver. Drug metabolism is a subset of general metabolism, focusing specifically on the transformation of xenobiotics (foreign substances).
Factors Affecting Drug Metabolism
Genetics: Genetic polymorphisms can lead to variability in enzyme activity, affecting drug efficacy and toxicity.
Physiology: Age, sex, and organ function (especially liver and kidney) influence metabolic rates.
Pharmacodynamics: Drug interactions and receptor sensitivity can alter metabolism.
Environment: Diet, exposure to chemicals, and lifestyle factors (e.g., smoking, alcohol) can induce or inhibit metabolic enzymes.
Phase I Reactions
Overview of Phase I Reactions
Phase I reactions introduce or expose functional groups on drugs, increasing their polarity and preparing them for further metabolism or excretion. These reactions are primarily catalyzed by enzymes such as cytochrome P450s, flavin monooxygenases, and dehydrogenases.
Oxidation: Addition of oxygen or removal of hydrogen.
Reduction: Addition of hydrogen or removal of oxygen.
Hydrolysis: Cleavage of bonds by the addition of water.
Deamination: Removal of an amino group.
Dealkylation: Removal of alkyl groups (e.g., methyl, ethyl).
Dehalogenation: Removal of halogen atoms (e.g., Cl, Br).
Cytochrome P450 (CYP) Enzymes
Structure and Function
Cytochrome P450 enzymes are a superfamily of heme-containing monooxygenases responsible for the oxidative metabolism of a wide variety of endogenous and exogenous compounds. They play a central role in drug metabolism, particularly in Phase I reactions.
CYP Families and Subfamilies
CYP enzymes are classified into families and subfamilies based on their amino acid sequence similarities. The main families involved in drug metabolism are CYP1, CYP2, and CYP3.
CYP1: Metabolizes polycyclic aromatic hydrocarbons, arylamines, nitrosamines, and caffeine. Generally inducible.
CYP2: Metabolizes approximately 50% of drugs. Usually constitutive, with some isoforms inducible. Includes CYP2C8, CYP2C9, CYP2C19, CYP2D6, and CYP2E1.
CYP3: Metabolizes approximately 35% of drugs. Usually inducible. Includes CYP3A4, CYP3A5, and CYP3A7.
CYPs and Endogenous vs. Xenobiotic Metabolism
Endogenous Substances: CYP7, CYP11, CYP17, CYP19, CYP21, CYP27 are involved in the synthesis and metabolism of steroids and other endogenous molecules.
Xenobiotic Substances: CYP1, CYP2, and CYP3 families are primarily responsible for the metabolism of drugs and other foreign compounds.
Examples of CYP-Mediated Metabolism
CYP1A1: Metabolizes polycyclic aromatic hydrocarbons.
CYP1A2: Metabolizes arylamines, nitrosamines, and caffeine.
CYP2C9: Metabolizes diazepam, phenytoin, and tolbutamide.
CYP2D6: Metabolizes lipophilic amines; inhibited by quinidine.
CYP2E1: Metabolizes halogenated hydrocarbons, ethanol, and benzene; induced in alcoholics.
CYP3A4: Metabolizes over 33% of drugs; highly inducible.
Other Phase I Enzymes
Flavin Monooxygenases (FMOs)
FMOs use flavin-adenine dinucleotide (FAD) to catalyze the oxygenation of nucleophilic nitrogen, sulfur, and phosphorus atoms in drugs. Major isoforms include FMO1, FMO2, and FMO5.
Alcohol and Aldehyde Dehydrogenases
Alcohol Dehydrogenases: Use NAD+ to oxidize primary alcohols to aldehydes and secondary alcohols to ketones.
Aldehyde Dehydrogenases: Use NAD+ to oxidize aldehydes to carboxylic acids.
Types of Oxidation Reactions
Aromatic and Alkene Oxidation
These reactions introduce hydroxyl groups into aromatic rings or alkenes, increasing polarity and facilitating further metabolism.
Benzylic, Allylic, and Aliphatic Oxidation
Oxidation at benzylic, allylic, or aliphatic positions leads to the formation of alcohols, aldehydes, or carboxylic acids, depending on the substrate and enzyme involved.
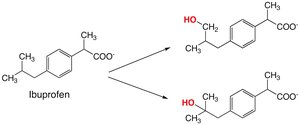
Oxidative Deamination and Dealkylation
Oxidative deamination removes amino groups, while oxidative dealkylation removes alkyl groups from nitrogen, oxygen, or sulfur atoms.
N-, O-, and S-Dealkylation and Oxidation
These reactions involve the removal of alkyl groups or the addition of oxygen to nitrogen, oxygen, or sulfur atoms, increasing drug polarity.
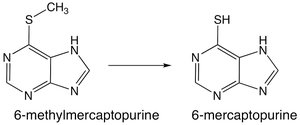
Dehalogenation
Dehalogenation reactions remove halogen atoms from drugs, often resulting in the formation of more polar metabolites.
Reduction and Hydrolysis
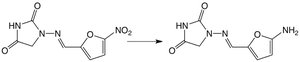
Reduction Reactions
Reduction reactions add hydrogen to or remove oxygen from drug molecules, often converting nitro groups to amines or carbonyls to alcohols.
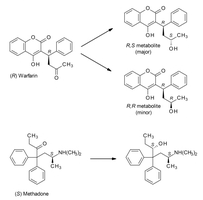
Hydrolysis Reactions
Hydrolysis involves the cleavage of chemical bonds by the addition of water, commonly affecting esters, amides, and other functional groups in drugs.
Summary Table: Major CYP Families and Their Roles
CYP Family | Inducibility | Main Substrates | Example Drugs |
|---|---|---|---|
CYP1 | Inducible | Polycyclic aromatic hydrocarbons, arylamines, nitrosamines, caffeine | CYP1A1, CYP1A2 |
CYP2 | Usually constitutive | ~50% of drugs | CYP2C9 (diazepam, phenytoin), CYP2D6 (lipophilic amines), CYP2E1 (ethanol) |
CYP3 | Inducible | ~35% of drugs | CYP3A4 (many drugs), CYP3A5, CYP3A7 |
Additional info: Drug metabolism is a critical aspect of pharmacology and toxicology, influencing drug efficacy, safety, and individual patient response. Understanding the enzymes and reactions involved is essential for predicting drug interactions and adverse effects.
