 Back
BackFluid, Electrolyte, and Acid-Base Homeostasis: ANP College Study Guide
Study Guide - Smart Notes

Fluid, Electrolyte, and Acid-Base Homeostasis
Body Fluids and Fluid Balance
Body fluids encompass all water-based liquids in the body, including blood plasma, interstitial fluid, cytosol, cerebrospinal fluid, lymph, and exocrine secretions. Water is the main component of these fluids. Fluid balance refers to maintaining the appropriate volume and concentration of intracellular and extracellular fluids, governed by the principle of mass balance: what is gained must equal what is lost.
Body Fluids: Include plasma, interstitial fluid, cytosol, and specialized fluids.
Fluid Balance: Ensures homeostasis by matching water input and output.
Principle of Mass Balance: Disturbances occur if the body cannot compensate for imbalances.
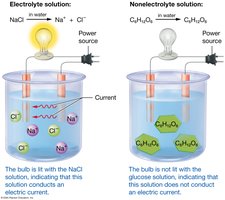
Electrolytes and Nonelectrolytes
Electrolytes are compounds with ionic bonds that dissociate into ions in water, enabling electrical conduction. Nonelectrolytes, with covalent bonds, do not dissociate and do not conduct electricity. Electrolyte balance is maintained by mass balance and is crucial for physiological function.
Electrolytes: Dissociate into ions; conduct electricity.
Nonelectrolytes: Do not dissociate; do not conduct electricity.
Electrolyte Balance: Maintained by equal intake and loss.
Acids, Bases, and pH
Acids release hydrogen ions in water, while bases accept hydrogen ions. Buffer systems resist dramatic swings in pH. The pH scale ranges from acidic (<7) to basic (>7), with 7 being neutral.
Acid: Releases H+ ions (e.g., HCl).
Base: Accepts H+ ions (e.g., NaOH, bicarbonate ion).
Buffer Systems: Bind H+ to stabilize pH.
pH Scale: Acidic <7, Basic >7, Neutral = 7.
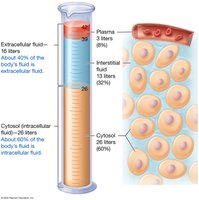
Total Body Water and Fluid Compartments
Total Body Water
Total body water varies by age, sex, body mass, and tissue composition. Muscle tissue contains more water than adipose tissue. Women generally have lower total body water than men. Infants have the highest percentage, while older adults have less.
Average Adult Male: ~42 liters (about 60% of body mass).
Factors: Age, sex, adipose vs. muscle tissue.
Fluid Compartments: Intracellular and Extracellular
The body’s fluids are divided into intracellular (ICF) and extracellular (ECF) compartments. ICF is the fluid within cells, while ECF includes plasma and interstitial fluid.
ICF: ~60% of total body water (cytosol).
ECF: Plasma (~8%), Interstitial fluid (~32%).
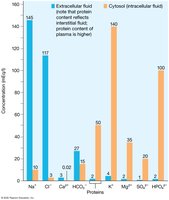
Solute Composition of Fluid Compartments
Solute concentrations differ between ECF and ICF. ECF is rich in sodium, chloride, calcium, and bicarbonate ions, while ICF contains more proteins, potassium, magnesium, sulfate, and monohydrogen phosphate ions.
ECF: High Na+, Cl-, Ca2+, HCO3-.
ICF: High K+, Mg2+, SO42-, HPO42-, proteins.
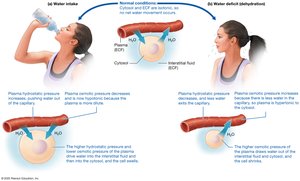
Movement of Water Between Compartments
Water moves freely between compartments, driven by hydrostatic and osmotic pressure gradients. Hydrostatic pressure pushes water out of capillaries, while osmotic pressure pulls water toward areas with higher solute concentration.
Hydrostatic Pressure: Force exerted by fluid; moves water from high to low pressure.
Osmotic Pressure: Moves water toward higher solute concentration (osmolarity).
Tonicity: Isotonic (equal osmotic pressure), hypotonic (cell swells), hypertonic (cell shrivels).
Water Losses and Gains
Water Losses
Water is lost through urine, feces, skin, and respiration. Obligatory water loss is the minimum required to excrete solutes. Sensible losses are noticed (urine, feces), while insensible losses are not (skin, respiration).
Obligatory Water Loss: ~500 ml/day (minimum urine output).
Sensible Loss: Urine (~1500 ml), feces (~100 ml).
Insensible Loss: Skin (~600 ml), respiration (~300 ml).
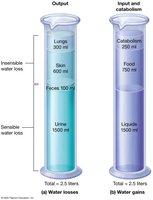
Water Gains
Water is gained from metabolic reactions, food, and liquid intake. The thirst mechanism, controlled by osmoreceptors in the hypothalamus, regulates intake.
Metabolic Water: ~250 ml/day.
Food: ~750 ml/day.
Liquids: ~1500 ml/day.
Thirst Mechanism: Triggered by increased plasma osmolarity or decreased plasma volume.
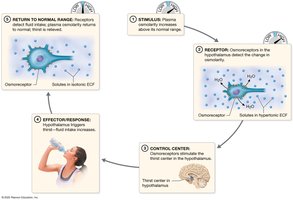
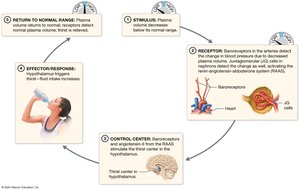
Hormonal Regulation of Fluid Balance
Four hormones regulate fluid balance: angiotensin II, aldosterone, atrial natriuretic peptide (ANP), and antidiuretic hormone (ADH). ADH is most important, increasing water reabsorption in the kidneys.
ADH: Increases water reabsorption, decreases urine output.
Angiotensin II & Aldosterone: Increase sodium and water retention.
ANP: Decreases sodium and water reabsorption.
Imbalances of Fluid Homeostasis
Dehydration
Dehydration is characterized by decreased ECF volume and increased osmolarity, often caused by sweating, diarrhea, vomiting, or diuretics. Symptoms include thirst, dry skin, oliguria, and can progress to organ failure.
Causes: Fluid loss, electrolyte imbalance.
Treatment: Moderate water intake or electrolyte-containing drinks.
Overhydration (Hypotonic Hydration)
Overhydration results in increased ECF volume and decreased osmolarity, causing cells to swell. It can lead to hyponatremia and cerebral edema.
Causes: Excess water intake, impaired renal function, ADH hypersecretion.
Treatment: Administration of hypertonic fluids.
Isosmotic Fluid Imbalances
Blood loss (hypovolemia) and fluid accumulation (hypervolemia) are isosmotic imbalances. Hypovolemia can lead to shock; hypervolemia causes edema.
Hypovolemia: Proportional loss of water and solutes.
Hypervolemia: Fluid accumulation without osmotic change.
Electrolyte Homeostasis
Sodium Ions
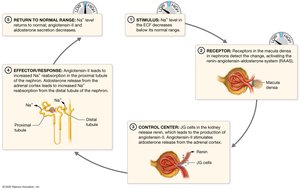
Sodium is the most abundant extracellular cation, essential for membrane potential and action potentials. Its concentration is regulated by the Na+/K+ ATPase pump and hormonal feedback loops.
Normal Range: 135–145 mEq/L.
Regulation: RAAS (renin-angiotensin-aldosterone system), ANP.
Imbalances: Hypernatremia (dehydration), hyponatremia (overhydration).
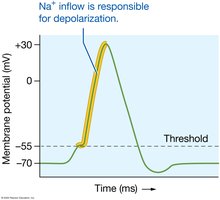
Potassium Ions
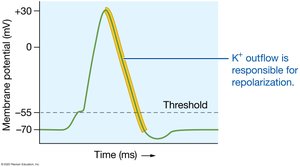
Potassium is the most abundant intracellular cation, crucial for repolarization of action potentials. Its concentration is regulated by the Na+/K+ ATPase pump, aldosterone, insulin, and epinephrine.
Normal Range: 3.9–4.5 mEq/L (ECF).
Imbalances: Hyperkalemia (renal failure, drug toxicity), hypokalemia (diuretics).
Calcium and Phosphate Ions
Calcium is required for muscle contraction, neuronal transmission, and blood clotting. Phosphate is integral to ATP and bone structure. Both are regulated by parathyroid hormone (PTH) and vitamin D3.
Calcium: 99% stored in bone; normal ECF range 8.7–10.4 mg/dL.
Phosphate: Found in hydroxyapatite crystals; regulated alongside calcium.
Imbalances: Hypercalcemia (neural depression), hypocalcemia (neural hyperexcitability).
Other Critical Ions
Chloride: High in ECF; involved in osmotic balance and HCl production.
Magnesium: Enzyme activation, bone tissue, acts as osmotic laxative if ingested in excess.
Acid-Base Homeostasis
Sources of Acids and Bases
Acids and bases are produced metabolically and ingested. Volatile acids (e.g., CO2) can be eliminated via lungs; fixed acids (e.g., lactic acid, uric acid) are eliminated by kidneys.
Metabolic Acids: Carbon dioxide, lactic acid, uric acid, ketone bodies.
Dietary Sources: Amino acids, fatty acids, citric acid, bicarbonate.
Chemical Buffer Systems
Buffer systems consist of a weak acid and its conjugate base, resisting large swings in pH. Major systems include carbonic acid–bicarbonate, phosphate, and protein buffers.
Carbonic Acid–Bicarbonate: Main ECF buffer.
Phosphate: Important in cytosol and kidney tubules.
Protein: Major intracellular buffer; hemoglobin buffers in erythrocytes.
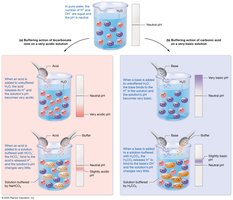
Physiological Buffer Systems: Respiratory and Renal Regulation
The respiratory system regulates volatile acids by controlling CO2 expiration. The urinary system excretes fixed acids and regulates bicarbonate ion concentration.
Respiratory System: Adjusts CO2 levels to match metabolic production.
Renal System: Secretes H+, reabsorbs HCO3-.
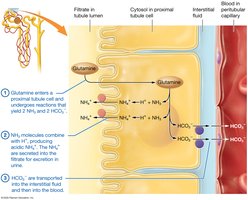
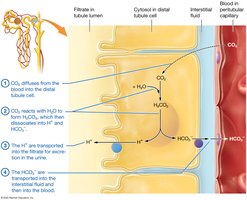
Acid-Base Imbalances
Acidosis and Alkalosis
Acidosis (pH < 7.35) and alkalosis (pH > 7.45) result from excess acids or bases. Compensation involves respiratory and renal adjustments, but permanent correction requires addressing the underlying cause.
Acidosis: Neuronal depression, coma, death.
Alkalosis: Neuronal hyperexcitability, seizures, death.
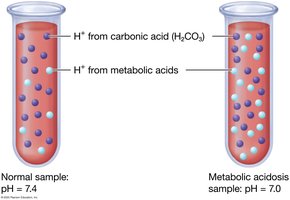
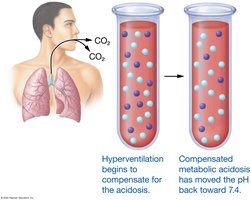
Dehydration: Example of Fluid, Electrolyte, and Acid-Base Imbalance
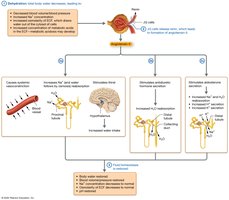
Dehydration leads to decreased blood volume, increased ECF osmolarity, and metabolic acidosis. Hormonal responses restore homeostasis by increasing water and sodium reabsorption and stimulating thirst.
Hormonal Response: Renin, angiotensin II, ADH, aldosterone.
Restoration: Water and sodium reabsorption, normalization of osmolarity and pH.
Summary Table: Acid-Base Disorders
Acid-base disorders are classified by their origin (respiratory or metabolic) and compensation status (uncompensated, partially, or fully compensated). Arterial blood gases (ABGs) are used to diagnose and monitor these disorders.
Disorder | pH | CO2 | HCO3- | Compensation |
|---|---|---|---|---|
Respiratory Acidosis | <7.35 | ↑ | ↑ (if compensated) | Renal |
Metabolic Acidosis | <7.35 | ↓ (if compensated) | ↓ | Respiratory |
Respiratory Alkalosis | >7.45 | ↓ | ↓ (if compensated) | Renal |
Metabolic Alkalosis | >7.45 | ↑ (if compensated) | ↑ | Respiratory |
Additional info: Table entries inferred from standard acid-base disorder classification.
