 Back
BackIntroduction to Physiology and Molecular Interactions: Foundational Concepts for ANP College Students
Study Guide - Smart Notes

Introduction to Physiology
Definition and Scope of Physiology
Physiology is the scientific study of the normal functions and processes of living organisms. It is distinct from anatomy, which focuses on the structure of organisms with less emphasis on function. Understanding physiology requires knowledge of the hierarchical organization of the human body, from atoms to the biosphere.
Anatomy: Study of structure.
Physiology: Study of function and processes.
Levels of Organization: Atoms → Molecules → Cells → Tissues → Organs → Organ Systems → Organism → Populations → Ecosystems → Biosphere.
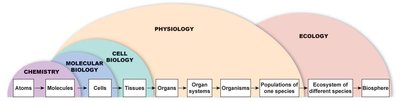
Review of Organ Systems
The human body is composed of ten major organ systems, each with specialized functions essential for survival and homeostasis.
Integumentary: Skin; forms a protective barrier.
Musculoskeletal: Support and movement.
Respiratory: Gas exchange.
Digestive: Nutrient and water uptake; waste elimination.
Urinary: Removal of excess water and waste.
Reproductive: Production of gametes (eggs or sperm).
Circulatory: Distribution of materials via blood.
Nervous and Endocrine: Coordination of body functions.
Immune: Protection from foreign invaders.
Function versus Process
Physiology distinguishes between the function (the "why" or teleological approach) and the process (the "how" or mechanistic approach) of biological phenomena. For example, red blood cells transport oxygen because cells need oxygen (function), and oxygen binds to hemoglobin in RBCs (process).
Homeostasis
Homeostasis is the body's ability to maintain a stable internal environment despite external changes. It involves the regulation of parameters such as osmolarity, temperature, pH, and the concentration of nutrients, water, ions, and hormones.
Controlled Parameters: Environmental factors (osmolarity, temperature, pH), materials for cells (nutrients, H2O, Na+, Ca2+, O2, hormones).
Scientific Inquiry and Experimental Design
Scientific inquiry in physiology relies on observation, hypothesis formation, experimentation, and analysis. Key concepts include:
Variables: Independent (manipulated) and dependent (measured).
Controls: Used for comparison.
Replication: Repeating experiments for reliability.
Placebo Effect: Positive psychological response to an inert treatment.
Nocebo Effect: Negative psychological response to an inert treatment.
Study Designs: Crossover, blind, double-blind, and double-blind crossover studies.
Molecular Interactions
Atoms, Ions, and Molecules
Atoms are the basic units of matter, composed of protons, neutrons, and electrons. The number of protons defines the element (atomic number), while the sum of protons and neutrons gives the atomic mass. Atoms can become ions (charged particles) or isotopes (same element, different number of neutrons).
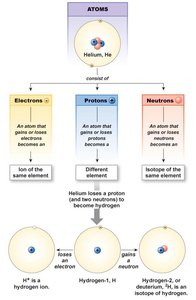
Types of Chemical Bonds
Chemical bonds are forces that hold atoms together in molecules and compounds. The main types include:
Covalent Bonds: Strong bonds formed by sharing electron pairs. Can be polar (unequal sharing, hydrophilic) or nonpolar (equal sharing, hydrophobic).
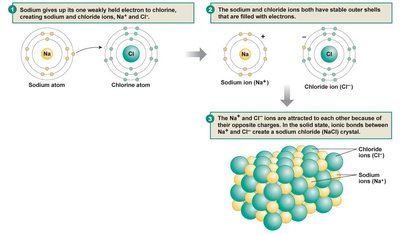
Ionic Bonds: Strong bonds formed by the transfer of electrons from one atom to another, resulting in attraction between oppositely charged ions.
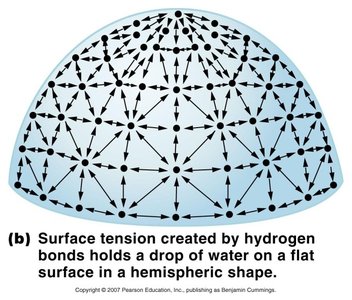
Hydrogen Bonds: Weak, partial bonds between a hydrogen atom and a nearby electronegative atom (F, O, or N). Responsible for water's surface tension and boiling/freezing points.
Van der Waals Forces: Weak, nonspecific attractions between atoms due to transient dipoles.
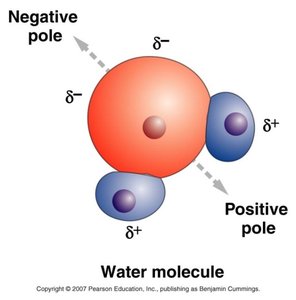
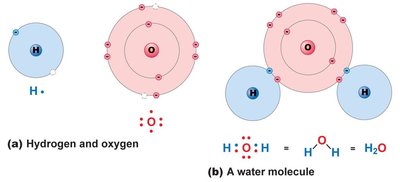
Molecular Shape and Function
The shape of a molecule, determined by the types of bonds and interactions, is closely related to its biological function. For example, the three-dimensional structure of proteins determines their activity and specificity.
Biomolecules
Biomolecules are organic molecules essential for life, classified into four main groups:
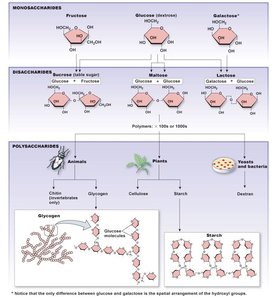
Carbohydrates: Composed of C, H, and O. Include monosaccharides (simple sugars), disaccharides, and polysaccharides (e.g., glycogen, starch).
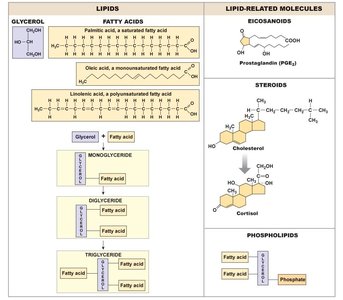
Lipids: Mostly C and H, nonpolar, not water-soluble. Include triglycerides, phospholipids, eicosanoids, and steroids (e.g., cholesterol).
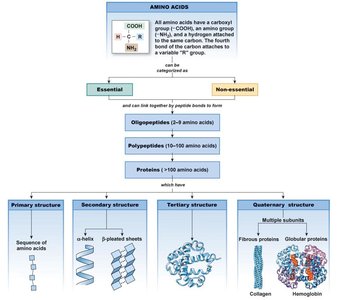
Proteins: Made of 20 amino acids (9 essential, 11 non-essential). Structure includes primary, secondary, tertiary, and quaternary levels. Most versatile biomolecules.
Nucleotides and Nucleic Acids: Composed of a base, sugar, and phosphate(s). Store and transmit genetic information (DNA, RNA) and participate in energy transfer (ATP, cAMP, NAD, FAD).
Combination Biomolecules
Some biomolecules are combinations of carbohydrates, proteins, and lipids:
Lipoproteins: Lipids + proteins; transport molecules in blood, component of cell membranes.
Glycoproteins: Carbohydrates + proteins; membrane structure.
Glycolipids: Carbohydrates + lipids; membrane receptors.
Aqueous Solutions and pH
The human body is approximately 60% water, with Na+, K+, and Cl– as the main ions in body fluids. Solutions are mixtures of solutes (dissolved substances) and solvents (liquids, usually water). The solubility of a molecule depends on its polarity: hydrophilic (polar/ionic) molecules dissolve easily, while hydrophobic (nonpolar) molecules do not.
Hydrogen Ion Concentration (pH)
The concentration of hydrogen ions (H+) determines the acidity of a solution, measured as pH. Maintaining pH homeostasis is critical because changes in pH can disrupt molecular bonds and protein function.
Acid: Contributes H+ to solution.
Base: Decreases H+ in solution.
pH Formula:
Buffers: Minimize changes in pH.

Summary Table: Key Concepts
Concept | Definition/Example |
|---|---|
Atom | Basic unit of matter (e.g., H, O, Na) |
Ion | Charged atom (e.g., Na+, Cl–) |
Isotope | Same element, different number of neutrons |
Covalent Bond | Shared electron pairs (e.g., H2O) |
Ionic Bond | Electron transfer, attraction of charges (e.g., NaCl) |
Hydrogen Bond | Weak, between H and F/O/N (e.g., water surface tension) |
Carbohydrate | Monosaccharides, disaccharides, polysaccharides |
Lipid | Triglycerides, steroids, phospholipids |
Protein | Amino acids, peptide bonds, structural levels |
Nucleotide | Base + sugar + phosphate; DNA, RNA, ATP |
Solution | Solute + solvent (e.g., salt water) |
pH | Acidity/alkalinity; |
