 Back
BackATP, Redox Reactions, and Cellular Respiration: Energy Flow in Cells
Study Guide - Smart Notes

ATP and Cellular Work
ATP: The Universal Energy Currency
ATP (adenosine triphosphate) is the primary energy carrier in all living cells. It powers nearly all forms of cellular work by providing energy through the hydrolysis of its high-energy phosphate bonds.
Structure: ATP consists of an adenine base, a ribose sugar, and three phosphate groups.
Function: ATP drives chemical, transport, and mechanical work in cells.
Hydrolysis: The reaction releases free energy ( kcal/mol), which is used to power cellular processes.
Regeneration: ATP is continuously regenerated from ADP and inorganic phosphate using energy derived from catabolic reactions (e.g., glucose breakdown).
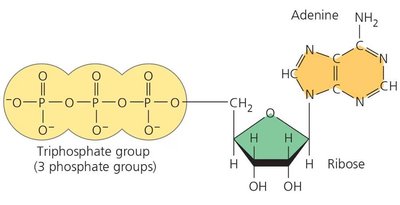
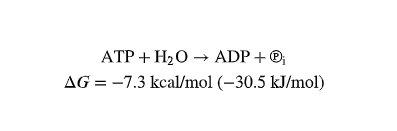
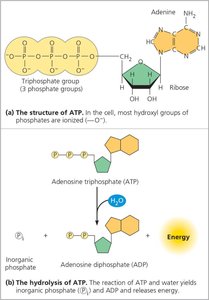
ATP Powers Cellular Work by Energy Coupling
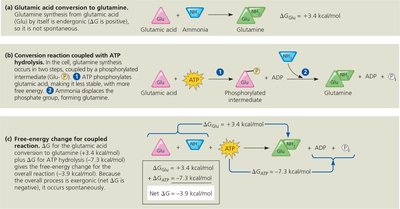
Cells couple exergonic (energy-releasing) reactions, such as ATP hydrolysis, to endergonic (energy-consuming) reactions. This process is called energy coupling and is essential for driving non-spontaneous cellular processes.
Phosphorylation: The transfer of a phosphate group from ATP to another molecule (protein or substrate) makes the recipient more reactive (a phosphorylated intermediate).
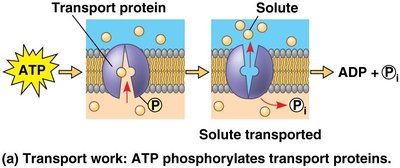
Examples: Protein activation, transport work (e.g., active transport across membranes), and mechanical work (e.g., muscle contraction).
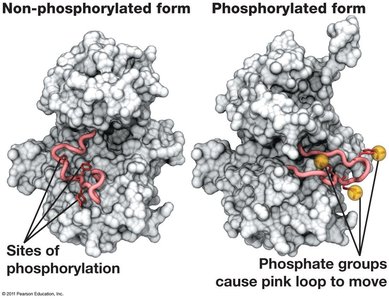
ATP as a Rechargeable Battery
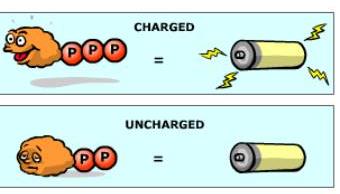
ATP is often compared to a rechargeable battery because it can be used, recharged, and used again. The energy to regenerate ATP from ADP comes from the breakdown of food molecules during cellular respiration.
Cycle: Energy from catabolism (exergonic reactions) is used to phosphorylate ADP, regenerating ATP, which is then used for cellular work (endergonic reactions).

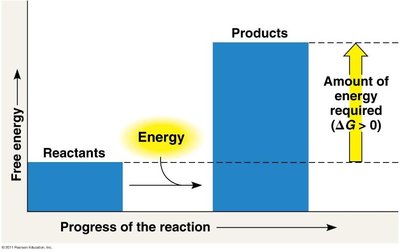
Exergonic and Endergonic Reactions
Energy Coupling in Cells
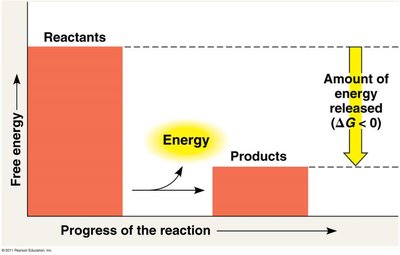
Cells use energy coupling to link exergonic and endergonic reactions, allowing energy released from one process to drive another that requires energy input.
Exergonic reactions: Release energy (), e.g., ATP hydrolysis.
Endergonic reactions: Require energy input (), e.g., synthesis of macromolecules.


Cellular Respiration and ATP Production
Overview of Cellular Respiration
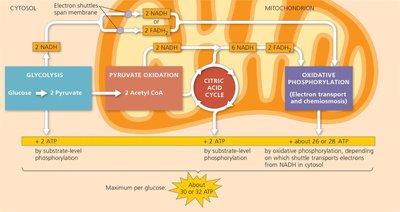
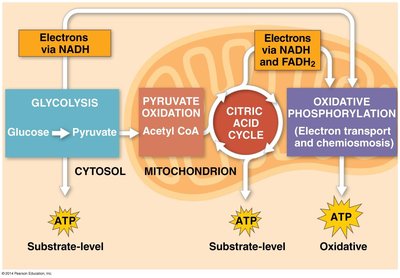
Cellular respiration is a series of metabolic pathways that convert the chemical energy in food into ATP. It involves glycolysis, pyruvate oxidation, the citric acid cycle, and oxidative phosphorylation (electron transport chain and chemiosmosis).
Aerobic respiration: Uses oxygen to maximize ATP yield.
Anaerobic respiration: Occurs without oxygen, yielding less ATP.
ATP yield: About 30–32 ATP molecules per glucose molecule.
ATP Synthesis Mechanisms
Substrate-level phosphorylation: Direct transfer of a phosphate group to ADP from a substrate (occurs in glycolysis and the citric acid cycle).
Oxidative phosphorylation: ATP synthesis powered by the electron transport chain and chemiosmosis (proton gradient across the mitochondrial membrane).

Redox Reactions in Cellular Respiration
Redox (Oxidation-Reduction) Reactions
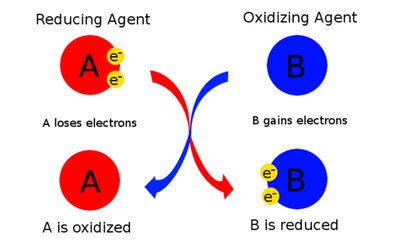
Redox reactions involve the transfer of electrons from one molecule (the reducing agent) to another (the oxidizing agent). These reactions are central to energy extraction from food molecules.
Oxidation: Loss of electrons (and usually hydrogen), resulting in decreased potential energy.
Reduction: Gain of electrons (and usually hydrogen), resulting in increased potential energy.
OIL RIG: Oxidation Is Loss, Reduction Is Gain (of electrons).
Electron carriers: Molecules like NAD+ and FAD shuttle electrons during cellular respiration, cycling between oxidized and reduced forms (NADH, FADH2).
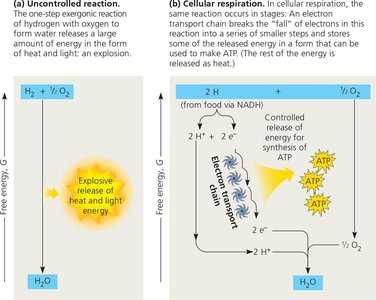


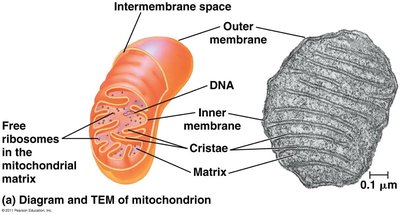
Electron Transport Chain and Chemiosmosis
The electron transport chain (ETC) is a series of protein complexes in the mitochondrial inner membrane that transfer electrons from NADH and FADH2 to oxygen, releasing energy used to pump protons and create a proton-motive force. This gradient drives ATP synthesis via ATP synthase (chemiosmosis).
Electron carriers: NADH and FADH2 donate electrons to the ETC.
ATP synthase: Enzyme that synthesizes ATP as protons flow back into the mitochondrial matrix.
Key Terms and Definitions
Term | Definition |
|---|---|
Adenosine Triphosphate (ATP) | Main energy currency of the cell |
Phosphorylation | Addition of a phosphate group to a molecule |
Redox Reaction | Reaction involving electron transfer (oxidation and reduction) |
Electron Carrier | Molecule that shuttles electrons (e.g., NAD+, FAD) |
Chemiosmosis | ATP generation using a proton gradient across a membrane |
Proton-motive force | Potential energy stored as a proton gradient |
ATP Synthase | Enzyme that synthesizes ATP using the proton gradient |
Oxidative Phosphorylation | ATP production using the electron transport chain and chemiosmosis |
Summary Table: Types of Cellular Work Powered by ATP
Type of Work | Example | ATP Role |
|---|---|---|
Chemical | Synthesis of macromolecules | Phosphorylation of reactants |
Transport | Active transport across membranes | Phosphorylation of transport proteins |
Mechanical | Muscle contraction, vesicle movement | ATP binds to motor proteins |
Additional info: This guide covers foundational concepts from Ch. 8 (An Introduction to Metabolism) and Ch. 9 (Cellular Respiration and Fermentation) of a college-level biology course, focusing on ATP, energy coupling, redox reactions, and the basics of cellular respiration.
