 Back
BackCellular Respiration and Fermentation: Mechanisms of Energy Harvest in Cells
Study Guide - Smart Notes

Cellular Respiration and Fermentation
Overview of Cellular Energy Transformation
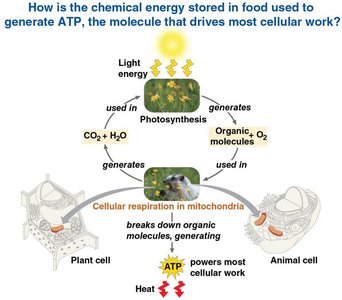
Cellular respiration and fermentation are essential metabolic pathways that allow cells to harvest energy from organic molecules. These processes convert the chemical energy stored in food into ATP, the primary energy currency of the cell, which powers most cellular work. Energy enters ecosystems as light and is transformed through photosynthesis and cellular respiration.
Photosynthesis uses CO2 and H2O to produce organic molecules and O2.
Cellular respiration uses O2 and organic molecules to generate ATP, releasing CO2 and H2O as waste.
Some energy is lost as heat during these processes.
Catabolic Pathways and ATP Production
Catabolic pathways break down complex molecules to release stored energy. The breakdown of organic molecules is exergonic, and the energy released is used to synthesize ATP.
Fermentation: Partial degradation of sugars without oxygen.
Aerobic respiration: Consumes organic molecules and oxygen, yielding ATP.
Anaerobic respiration: Similar to aerobic, but uses compounds other than oxygen as electron acceptors.
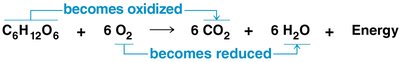
The overall equation for aerobic cellular respiration is:
Redox Reactions: Oxidation and Reduction
Principles of Redox Reactions
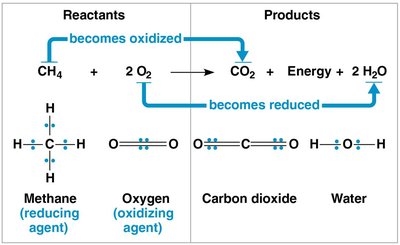
Redox reactions involve the transfer of electrons between reactants, releasing energy stored in organic molecules. These reactions are fundamental to cellular respiration.
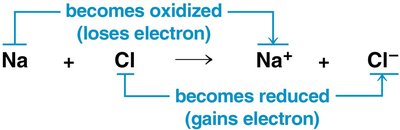
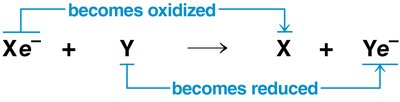
Oxidation: Loss of electrons from a substance.
Reduction: Gain of electrons by a substance (reduces positive charge).
Oxidation and reduction always occur together; the electron donor is the reducing agent, and the electron acceptor is the oxidizing agent.
Energy Release in Redox Reactions
Electrons lose potential energy when transferred from less electronegative atoms to more electronegative ones, such as oxygen. This energy is harnessed to synthesize ATP.
Organic molecules rich in hydrogen are excellent sources of high-energy electrons.
Cellular respiration is a redox process; energy is released as hydrogen and electrons are transferred to oxygen atoms.
Stepwise Energy Harvest: NAD+ and the Electron Transport Chain
Role of Electron Carriers
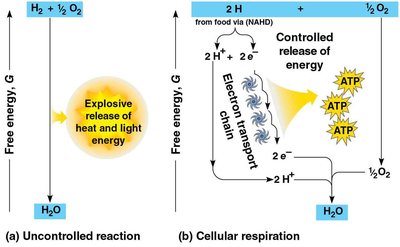
Glucose and other organic molecules are oxidized in a series of steps. Electrons are transferred to electron carriers, such as NAD+, which functions as an oxidizing agent.
NAD+ is reduced to NADH, storing energy for ATP synthesis.
If NADH transferred electrons directly to oxygen, energy would be released explosively; instead, the electron transport chain breaks the fall of electrons into manageable steps.

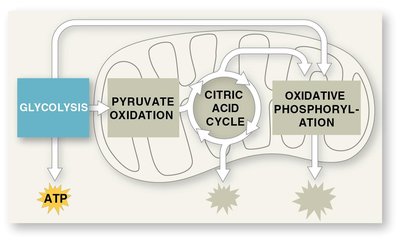
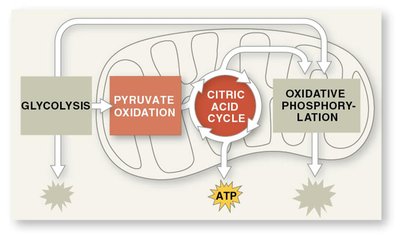
Stages of Cellular Respiration
Three Main Stages
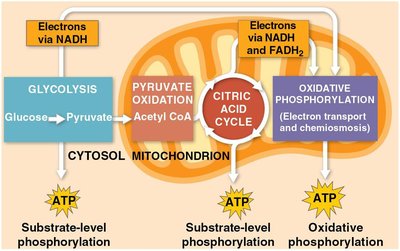
Cellular respiration consists of three main stages, each contributing to the breakdown of glucose and the production of ATP.
Glycolysis: Breaks down glucose into two molecules of pyruvate.
Pyruvate Oxidation and Citric Acid Cycle: Completes the breakdown of glucose to CO2.
Oxidative Phosphorylation: Electron transport chain and chemiosmosis facilitate synthesis of most ATP.

ATP Formation Mechanisms
Oxidative phosphorylation: Generates ~90% of ATP, powered by redox reactions.
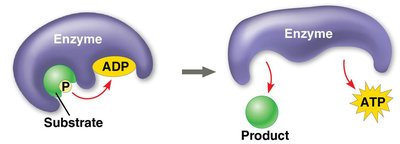
Substrate-level phosphorylation: Enzyme transfers a phosphate group directly from a substrate to ADP.
Glycolysis: Harvesting Energy from Glucose
Phases of Glycolysis
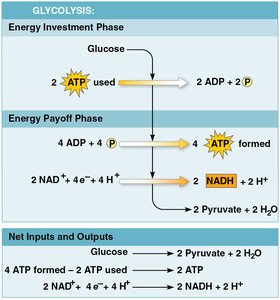
Glycolysis occurs in the cytoplasm and consists of two major phases:
Energy investment phase: 2 ATP are used to split glucose into two three-carbon sugar molecules.
Energy payoff phase: 4 ATP are synthesized, 2 NAD+ are reduced to NADH, and the sugars are oxidized to form 2 pyruvate and 2 H2O.
Net yield: 2 ATP by substrate-level phosphorylation.
Key Features of Glycolysis
All carbon from glucose is accounted for in pyruvate.
No CO2 is released; glycolysis occurs with or without oxygen.
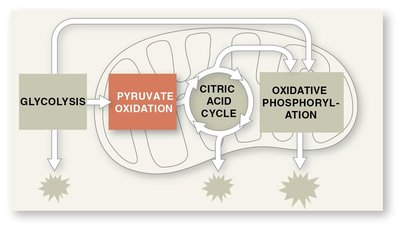
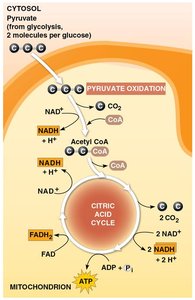
Pyruvate Oxidation and the Citric Acid Cycle
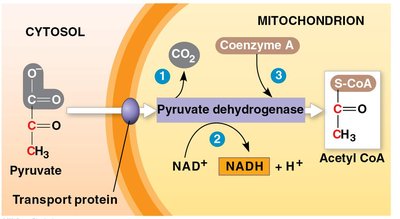
Oxidation of Pyruvate
Pyruvate is converted to acetyl coenzyme A (acetyl CoA) before entering the citric acid cycle. Pyruvate dehydrogenase catalyzes three reactions:
Oxidation of pyruvate’s carboxyl group, releasing CO2.
Reduction of NAD+ to NADH.
Combination of the remaining two-carbon fragment with coenzyme A to form acetyl CoA.
The Citric Acid Cycle (Krebs Cycle)
The citric acid cycle oxidizes organic fuel derived from pyruvate, generating ATP, NADH, and FADH2 per turn. The cycle runs twice per glucose molecule.
Eight steps, each catalyzed by a specific enzyme.
Acetyl group joins with oxaloacetate to form citrate; subsequent steps regenerate oxaloacetate.
NADH and FADH2 carry electrons to the electron transport chain.
Oxidative Phosphorylation: Electron Transport and Chemiosmosis
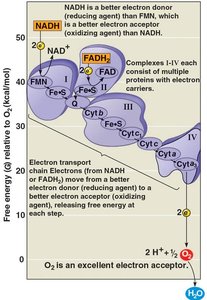
Electron Transport Chain
The electron transport chain is embedded in the inner mitochondrial membrane (or plasma membrane in prokaryotes). It consists of multiprotein complexes and carrier molecules, including cytochromes.
NADH and FADH2 donate electrons to the chain.
Electrons are passed through carriers, alternating between reduced and oxidized states.
Electrons drop in free energy, finally passing to O2 to form H2O.
No ATP is produced directly by the chain.
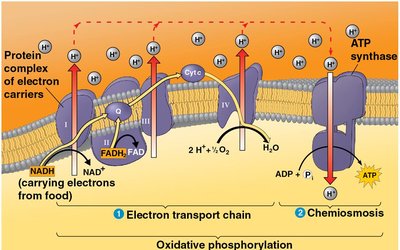
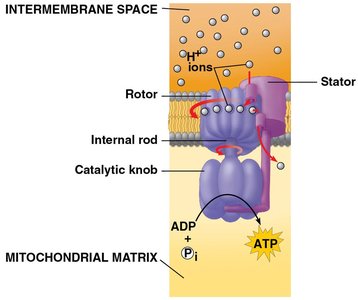
Chemiosmosis and ATP Synthase
Energy released by the electron transport chain is used to pump H+ ions across the membrane, creating a gradient. H+ flows back through ATP synthase, driving ATP production.
Chemiosmosis: Use of energy in a H+ gradient to drive cellular work.
ATP synthase acts as a molecular mill, catalyzing phosphorylation of ADP to ATP.
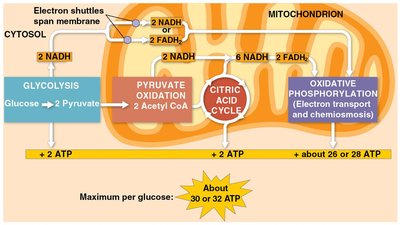
ATP Yield from Cellular Respiration
Energy Flow and ATP Production
Most energy flows in the sequence: glucose → NADH → electron transport chain → proton-motive force → ATP. About 34% of the energy in glucose is transferred to ATP, producing up to 32 ATP molecules per glucose.
Fermentation and Anaerobic Respiration
ATP Production Without Oxygen
When oxygen is unavailable, cells use fermentation or anaerobic respiration to produce ATP. Anaerobic respiration uses an electron transport chain with a final electron acceptor other than oxygen (e.g., sulfate ion).
Fermentation regenerates NAD+ by alternate mechanisms.
Types of Fermentation
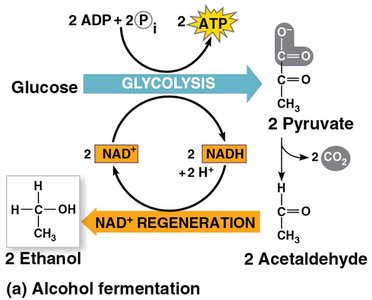
Alcohol fermentation: Pyruvate is converted to ethanol in two steps.
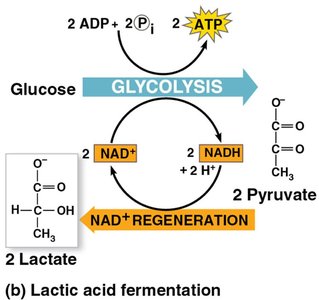
Lactic acid fermentation: Pyruvate is reduced directly to lactate.
Summary Table: Stages of Cellular Respiration
Stage | Main Location | Key Products | ATP Yield |
|---|---|---|---|
Glycolysis | Cytoplasm | Pyruvate, NADH | 2 ATP |
Pyruvate Oxidation | Mitochondrial Matrix | Acetyl CoA, NADH, CO2 | 0 ATP |
Citric Acid Cycle | Mitochondrial Matrix | NADH, FADH2, CO2 | 2 ATP |
Oxidative Phosphorylation | Inner Mitochondrial Membrane | ATP, H2O | ~28 ATP |
Fermentation | Cytoplasm | Ethanol or Lactate, NAD+ | 2 ATP |
Additional info: The above table summarizes the main stages, locations, products, and ATP yield of cellular respiration and fermentation, providing a concise comparison for exam preparation.
