 Back
BackCellular Respiration: Oxidative Phosphorylation, Anaerobic Respiration, and Fermentation
Study Guide - Smart Notes

Cellular Respiration Overview
Introduction to Cellular Respiration
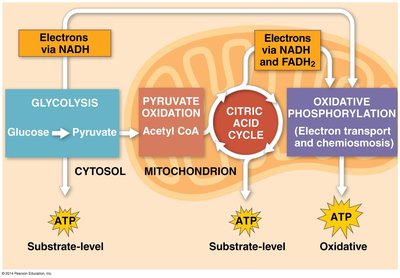
Cellular respiration is a series of metabolic processes by which cells convert biochemical energy from nutrients into adenosine triphosphate (ATP), releasing waste products. It consists of glycolysis, pyruvate oxidation, the citric acid cycle, and oxidative phosphorylation. This process is essential for the production of ATP, the energy currency of the cell.
ATP (Adenosine Triphosphate): The main energy carrier in cells.
Redox Reactions: Chemical reactions involving the transfer of electrons, crucial for energy extraction.
Electron Carriers: Molecules such as NADH and FADH2 that transport electrons to the electron transport chain.
Glycolysis
Key Features of Glycolysis
Glycolysis is the first stage of cellular respiration, occurring in the cytoplasm. It breaks down glucose into two molecules of pyruvate, generating a small amount of ATP and NADH. This process does not require oxygen (anaerobic).
Starting molecule: Glucose
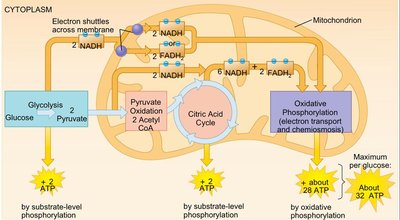
End products: 2 Pyruvate, 2 NADH, 2 H2O
ATP production: Net gain of 2 ATP per glucose
Location: Cytoplasm
Phases: Energy investment and energy payoff
Key steps: Phosphorylation, regulation (phosphofructokinase), splitting, redox reactions
Pyruvate Oxidation
Key Features of Pyruvate Oxidation
Pyruvate oxidation links glycolysis to the citric acid cycle. Each pyruvate is converted into acetyl-CoA, producing NADH and releasing CO2. This step occurs in the mitochondrion and does not directly produce ATP.
Reactants: Pyruvate, NAD+, Coenzyme A
End products: Acetyl-CoA, NADH, CO2
Location: Mitochondrial matrix
ATP production: None directly
NADH production: 1 NADH per pyruvate
Key steps: Decarboxylation, redox, acetyl-CoA formation
Citric Acid Cycle (Krebs Cycle)
Key Features of the Citric Acid Cycle
The citric acid cycle completes the oxidation of glucose derivatives, generating ATP, NADH, and FADH2. It occurs in the mitochondrial matrix and is a central hub for metabolic pathways.
Reactants: Acetyl-CoA, oxaloacetate
End products (per cycle): 1 ATP, 3 NADH, 1 FADH2, 2 CO2, oxaloacetate (regenerated)
Location: Mitochondrial matrix
ATP production: 1 ATP per cycle (substrate-level phosphorylation)
Key steps: Carbon dioxide release, redox reactions, regeneration of oxaloacetate
Oxidative Phosphorylation
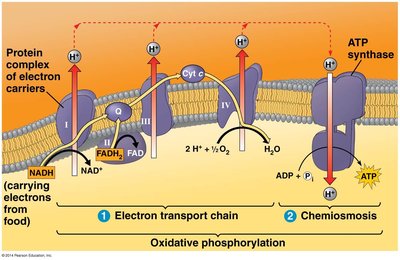
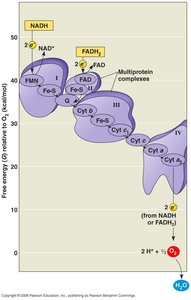
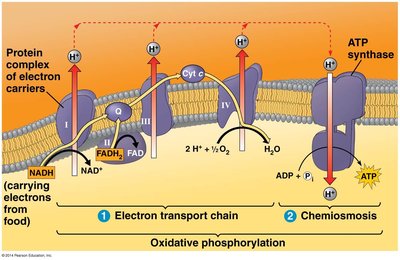
Electron Transport Chain (ETC)
Oxidative phosphorylation is the final stage of cellular respiration, consisting of the electron transport chain and chemiosmosis. NADH and FADH2 donate electrons to the ETC, a series of protein complexes in the inner mitochondrial membrane. The energy released pumps protons (H+) into the intermembrane space, creating a proton gradient.
Starting molecules: NADH, FADH2
End products: H2O, NAD+, FAD, H+ gradient
Location: Inner mitochondrial membrane
ATP production: None directly in ETC
Oxygen dependence: Aerobic (O2 is the final electron acceptor)
Key steps: Electron transfer, proton pumping, water formation
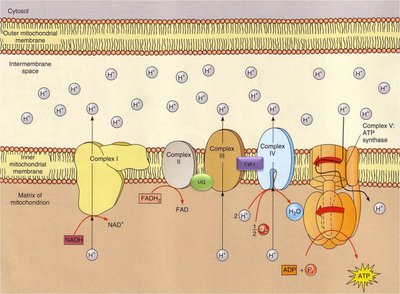
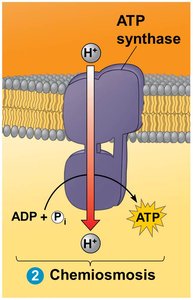
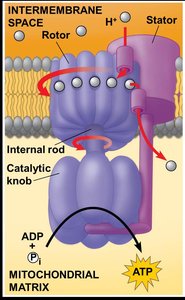
Chemiosmosis and ATP Synthase
The proton gradient generated by the ETC drives protons back into the mitochondrial matrix through ATP synthase, a molecular machine that synthesizes ATP from ADP and inorganic phosphate. This process is called chemiosmosis.
Starting molecules: H+ gradient
End products: 26–28 ATP per glucose
Location: Across the inner mitochondrial membrane
Oxygen dependence: Aerobic (relies on ETC)
Key steps: H+ flow through ATP synthase, ATP production
ATP Yield from Cellular Respiration
Summary Table of ATP Production
Stage | ATP Produced (per glucose) | Mechanism |
|---|---|---|
Glycolysis | 2 | Substrate-level phosphorylation |
Pyruvate Oxidation | 0 | — |
Citric Acid Cycle | 2 | Substrate-level phosphorylation |
Oxidative Phosphorylation | 26–28 | Oxidative phosphorylation |
Total | 30–32 | — |
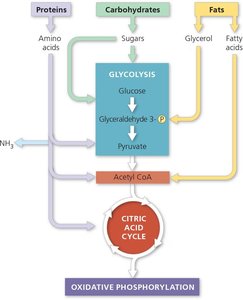
Metabolic Versatility and Catabolism of Other Molecules
Entry Points for Other Nutrients
Cellular respiration is a versatile pathway. Monomers from proteins, carbohydrates, and fats can enter at various points, after appropriate modifications (e.g., deamination of amino acids, β-oxidation of fatty acids).
Proteins: Deaminated and enter as pyruvate, acetyl-CoA, or citric acid cycle intermediates
Fats: Glycerol enters glycolysis; fatty acids enter as acetyl-CoA
Carbohydrates: Enter as glucose or glycolytic intermediates
Anaerobic Respiration and Fermentation
Fermentation
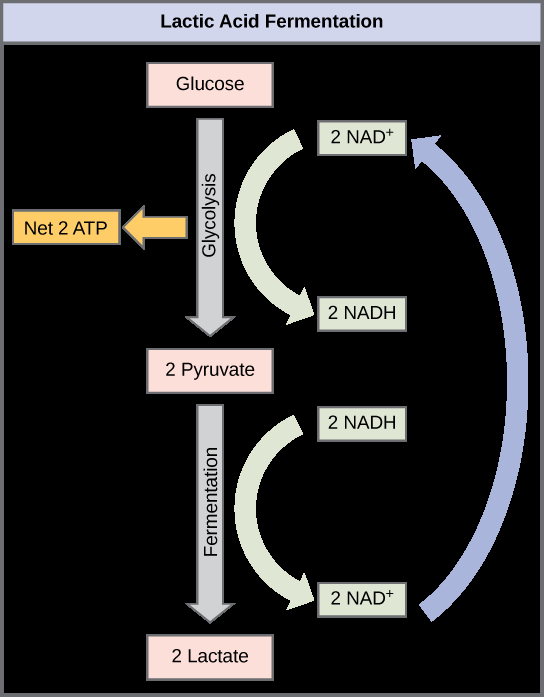
When oxygen is unavailable, cells can generate ATP through fermentation. Fermentation allows glycolysis to continue by regenerating NAD+ from NADH. There are two main types: lactic acid fermentation and alcohol fermentation.
Lactic Acid Fermentation: Pyruvate is reduced to lactic acid; occurs in muscle cells and some bacteria.
Alcohol Fermentation: Pyruvate is converted to ethanol and CO2; occurs in yeast and some bacteria.
ATP yield: Only 2 ATP per glucose (from glycolysis)
Electron acceptor: Organic molecule (e.g., pyruvate or acetaldehyde)
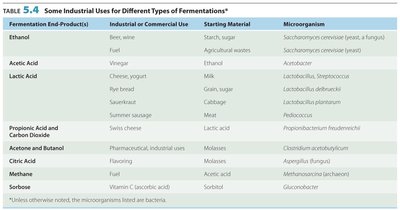
Industrial Uses of Fermentation
Fermentation is used in various industries to produce food, beverages, and chemicals. Different microorganisms and substrates yield different products.
Fermentation End-Product(s) | Industrial/Commercial Use | Starting Material | Microorganism |
|---|---|---|---|
Ethanol | Beer, wine, fuel | Starch, sugar | Saccharomyces cerevisiae |
Lactic Acid | Cheese, yogurt | Milk | Lactobacillus |
Acetic Acid | Vinegar | Alcohol | Acetobacter |
Methane | Fuel | Acetic acid | Methanobacterium |
Propionic Acid & CO2 | Swiss cheese | Lactic acid | Propionibacterium |
Acetone & Butanol | Pharmaceutical, industrial uses | Molasses | Clostridium acetobutylicum |
Citric Acid | Food additive | Molasses | Aspergillus niger |
Key Terminologies
Cellular respiration: The process of converting biochemical energy from nutrients into ATP.
Glycolysis: The breakdown of glucose to pyruvate, producing ATP and NADH.
Pyruvate oxidation: Conversion of pyruvate to acetyl-CoA, producing NADH and CO2.
Citric acid cycle: Series of reactions that generate ATP, NADH, and FADH2 from acetyl-CoA.
ATP (Adenosine Triphosphate): Main energy currency of the cell.
Phosphorylation: Addition of a phosphate group to a molecule.
Redox reactions: Chemical reactions involving electron transfer.
Electron carrier molecules: Molecules that transport electrons (e.g., NADH, FADH2).
NADH (Nicotinamide adenine dinucleotide): Electron carrier in cellular respiration.
Chemiosmosis: Process by which ATP is produced using the energy of a proton gradient.
Proton-motive force: The force generated by the transmembrane proton gradient.
ATP Synthase: Enzyme that synthesizes ATP using the proton gradient.
Oxidative phosphorylation: Production of ATP using energy derived from the redox reactions of the electron transport chain.
