 Back
BackMetabolism, Energy, and Enzymes: Foundations of Cellular Biochemistry
Study Guide - Smart Notes

Introduction to Metabolism
Overview of Metabolic Pathways
Metabolism encompasses all the chemical reactions occurring within a cell, divided into two main types: catabolic and anabolic pathways. These pathways are essential for maintaining cellular structure, function, and energy balance.
Catabolism: The breakdown of large molecules into smaller units, releasing energy. Example: Hydrolysis of proteins into amino acids.
Anabolism: The synthesis of larger molecules from smaller ones, requiring energy input. Example: Formation of proteins from amino acids.
Metabolic pathways: Serve as blueprints for all cellular reactions, integrating catabolic and anabolic processes.
Key concept: In living systems, mass/matter and energy are conserved; they are transformed, not created or destroyed.
Example: The breakdown of glucose (a carbohydrate) in catabolic pathways releases energy for cellular work.
Conservation of Matter and Energy
Principles of Conservation
Biological reactions obey the laws of conservation of matter and energy. Atoms present in reactants are conserved in products, and energy is transformed between different forms.
Law of Conservation of Matter: Atoms are neither created nor destroyed in chemical reactions.
Law of Conservation of Energy: Energy is not created or destroyed, only converted (e.g., chemical energy to heat).
Example: If a reaction starts with 7 carbon atoms in the reactants, the products will also contain 7 carbon atoms.
Energy in Biological Systems
Types of Energy
Cells utilize various forms of energy to perform work and drive reactions.
Kinetic energy: Energy of motion (e.g., movement of molecules).
Potential energy: Energy stored due to position or structure (e.g., chemical bonds, concentration gradients).
Chemical energy: Energy stored in molecular bonds, released during catabolic reactions.
Free energy (G): The portion of a system's energy available to do work at constant temperature and pressure.
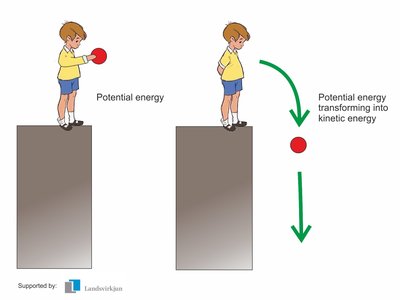
Example: The conversion of chemical potential energy in glucose to kinetic energy during muscle contraction.
Spontaneity and Free Energy Change (ΔG)
Determining Reaction Spontaneity
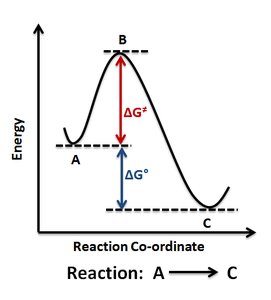
The spontaneity of a reaction is determined by the change in free energy (ΔG). This value predicts whether a reaction will occur without external energy input.
ΔG = Gproducts – Greactants
Exergonic reactions: ΔG is negative; energy is released; reaction is spontaneous.
Endergonic reactions: ΔG is positive; energy is absorbed; reaction is non-spontaneous.
Activation energy (EA): The initial energy required to start a reaction, represented as a 'hump' in energy diagrams.
Example: ATP hydrolysis is an exergonic reaction that releases energy to drive endergonic processes in the cell.
Coupling of Reactions in Cells
Pairing Exergonic and Endergonic Reactions
Cells often couple exergonic reactions (such as ATP hydrolysis) with endergonic reactions to drive essential processes that would not occur spontaneously.
ATP hydrolysis: ATP → ADP + Pi; ΔG = -7.3 kcal/mol
Coupling: The energy released from ATP hydrolysis is used to power endergonic reactions, such as synthesis of complex molecules.
Example: Synthesis of glutamine from glutamic acid and ammonia is endergonic, but becomes exergonic when paired with ATP hydrolysis.

Additional info: Coupling is fundamental to cellular metabolism, enabling cells to perform work and maintain homeostasis.
Activation Energy and Reaction Rates
Role of Activation Energy
Activation energy is the barrier that must be overcome for a reaction to proceed. Lower activation energy increases reaction rates.
Source: Usually provided by heat, causing molecular collisions.
Biological challenge: Most cellular reactions require activation energies not achievable at physiological temperatures.
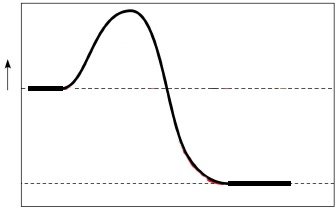
Example: Burning wood requires activation energy from a match; in cells, enzymes provide an alternative.
Enzymes: Biological Catalysts
Structure and Function of Enzymes
Enzymes are proteins (or RNA) that act as catalysts, speeding up reactions by lowering activation energy without altering ΔG.
Active site: The region where substrates bind and reactions occur.
Substrate specificity: Enzymes are highly specific, often named with the suffix '-ase'.
Mechanisms: Enzymes orient substrates, strain bonds, provide favorable environments, and may form temporary covalent bonds.
Enzyme activity: Influenced by temperature and pH; denaturation occurs if conditions are extreme.
Example: HIV protease catalyzes the cleavage of peptide bonds in viral proteins.
Enzyme Regulation and Environmental Effects
Factors Affecting Enzyme Activity
Enzyme function is optimized for the conditions in which the organism evolved. Activity increases with temperature up to a point, after which denaturation occurs.
Temperature: Higher temperatures increase activity until denaturation.
pH: Each enzyme has an optimal pH; extreme pH can denature the enzyme.
Example: Enzymes from E. coli (gut bacterium) and Great Salt Lake bacteria differ in salt tolerance.
Summary Table: Exergonic vs. Endergonic Reactions
Type of Reaction | ΔG | Energy Flow | Spontaneity | Example |
|---|---|---|---|---|
Exergonic | Negative | Energy released | Spontaneous | ATP hydrolysis |
Endergonic | Positive | Energy absorbed | Non-spontaneous | Protein synthesis |
Key Equations
Conclusion
Metabolism, energy transformations, and enzyme function are central to cellular life. Understanding these concepts is essential for studying cell biology, biochemistry, and physiology.
