 Back
BackCh. 10: Aerobic Respiration-Cellular Energy Metabolism and Mitochondrial Function
Study Guide - Smart Notes

Aerobic Respiration and Cellular Energy Metabolism
Introduction to Aerobic Respiration
Aerobic respiration is a multi-step metabolic pathway by which cells convert biochemical energy from nutrients into adenosine triphosphate (ATP), using oxygen as the final electron acceptor. This process is essential for the survival of eukaryotic cells and occurs primarily within the mitochondria.
Definition: Aerobic respiration involves the oxidation of organic molecules, with the transfer of electrons through a series of carriers, ultimately reducing oxygen to water.
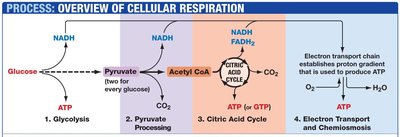
Main Stages: Glycolysis, Pyruvate Oxidation, Citric Acid Cycle (Krebs Cycle), and Electron Transport Chain with Chemiosmotic ATP Synthesis.
ATP Production: The majority of ATP is generated during the electron transport and chemiosmotic coupling stages.
Mitochondrial Structure and Function
Mitochondrial Anatomy
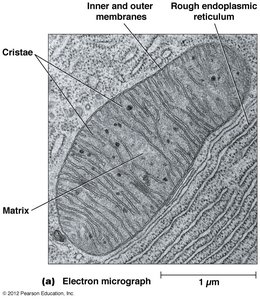
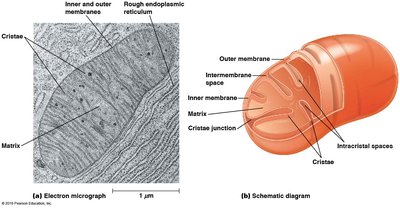
Mitochondria are double-membraned organelles that serve as the site of aerobic respiration. Their structure is highly specialized to facilitate efficient energy conversion.
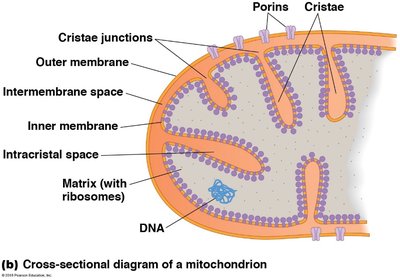
Outer Membrane: Contains porins for the passage of ions and small molecules.
Inner Membrane: Highly folded into cristae, houses the electron transport chain and ATP synthase.
Matrix: Contains enzymes for the citric acid cycle, mitochondrial DNA, and ribosomes.
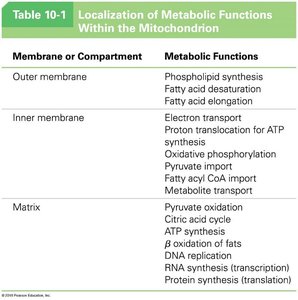
Localization of Metabolic Functions
The compartmentalization within mitochondria allows for the segregation and regulation of metabolic pathways.
Membrane or Compartment | Metabolic Functions |
|---|---|
Outer membrane | Phospholipid synthesis, fatty acid desaturation, fatty acid elongation |
Inner membrane | Electron transport, proton translocation for ATP synthesis, oxidative phosphorylation, pyruvate import, fatty acyl CoA import, metabolite transport |
Matrix | Pyruvate oxidation, citric acid cycle, ATP synthesis, β oxidation of fats, DNA replication, RNA synthesis (transcription), protein synthesis (translation) |
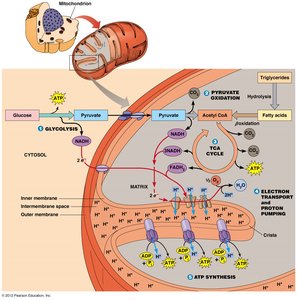
Major Steps of Aerobic Respiration
1. Glycolysis
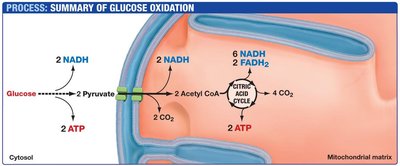
Glycolysis occurs in the cytoplasm and breaks down glucose into two molecules of pyruvate, generating a net gain of 2 ATP and 2 NADH per glucose molecule.
Location: Cytoplasm
Products: 2 Pyruvate, 2 NADH, 2 ATP
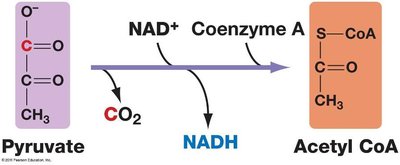
2. Pyruvate Oxidation
Pyruvate is transported into the mitochondrial matrix, where it is converted into acetyl CoA by the pyruvate dehydrogenase complex. This reaction produces NADH and releases CO2.
Enzyme Complex: Pyruvate dehydrogenase (multi-enzyme complex with 3 enzymes, 5 coenzymes, and 2 regulatory proteins)
Reaction: Pyruvate + NAD+ + CoA → Acetyl CoA + NADH + CO2
3. Citric Acid Cycle (Krebs Cycle)
The citric acid cycle, also known as the tricarboxylic acid (TCA) cycle or Krebs cycle, completes the oxidation of acetyl CoA to CO2. It generates NADH, FADH2, and GTP (or ATP).
Location: Mitochondrial matrix
Products per Acetyl CoA: 3 NADH, 1 FADH2, 1 GTP (or ATP), 2 CO2
Key Steps: Decarboxylation, oxidation, substrate-level phosphorylation, regeneration of oxaloacetate
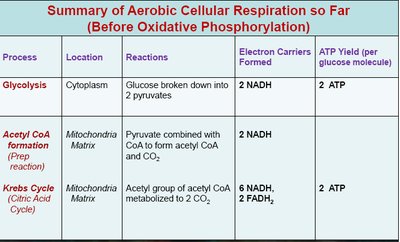
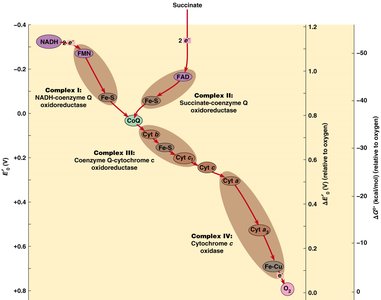
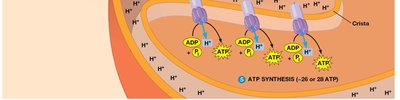
4. Electron Transport Chain (ETC) and Chemiosmosis
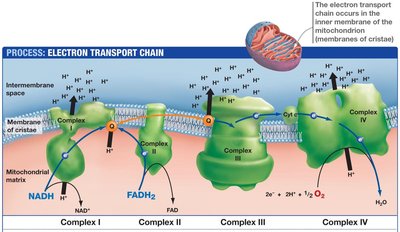
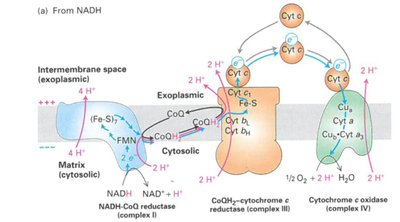
The ETC is a series of protein complexes embedded in the inner mitochondrial membrane. Electrons from NADH and FADH2 are transferred through these complexes to oxygen, the terminal electron acceptor, forming water. The energy released is used to pump protons across the membrane, creating a proton gradient.
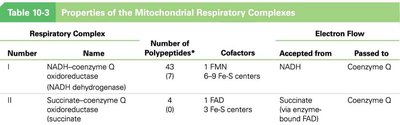
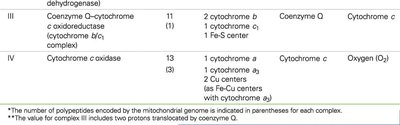
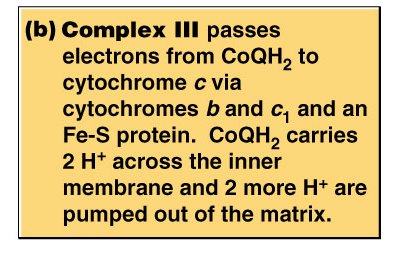
Complexes: I (NADH-CoQ oxidoreductase), II (Succinate-CoQ oxidoreductase), III (CoQ-cytochrome c oxidoreductase), IV (Cytochrome c oxidase)
Electron Carriers: Flavoproteins, iron-sulfur proteins, cytochromes, coenzyme Q (ubiquinone), copper-containing cytochromes
Proton Motive Force: The electrochemical gradient drives ATP synthesis via ATP synthase (F0F1 ATPase)
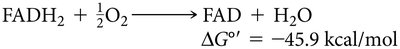



ATP Synthesis and the Chemiosmotic Mechanism
ATP synthase utilizes the proton motive force generated by the ETC to synthesize ATP from ADP and inorganic phosphate. The binding change model describes the conformational changes in ATP synthase subunits that facilitate ATP production.
Binding Change Model: β subunits of ATP synthase cycle through loose (L), tight (T), and open (O) conformations to bind substrates, catalyze ATP formation, and release ATP.
Energy Source: The energy for ATP synthesis is provided by the flow of protons down their electrochemical gradient.
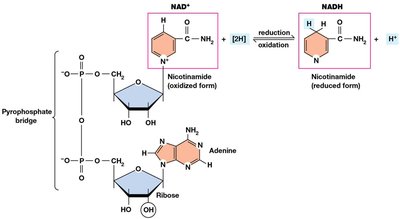
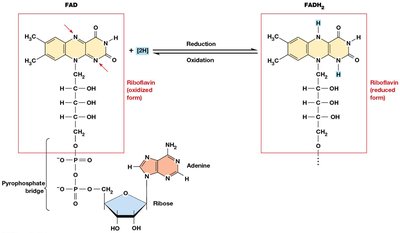
Electron Carriers: NAD+ and FAD
Structure and Function
NAD+ (nicotinamide adenine dinucleotide) and FAD (flavin adenine dinucleotide) are essential coenzymes that function as electron carriers in cellular respiration.
NAD+: Accepts two electrons and one proton to become NADH.
FAD: Accepts two electrons and two protons to become FADH2.
Summary of ATP Yield
The complete aerobic oxidation of one molecule of glucose yields a maximum theoretical value of approximately 30–32 ATP, depending on the shuttle systems and cell type.
Glycolysis: 2 ATP (net), 2 NADH
Pyruvate Oxidation: 2 NADH
Krebs Cycle: 2 ATP (or GTP), 6 NADH, 2 FADH2
Electron Transport and Chemiosmosis: Each NADH yields ~2.5 ATP, each FADH2 yields ~1.5 ATP
Process | Location | Reactions | Electron Carriers Formed | ATP Yield (per glucose molecule) |
|---|---|---|---|---|
Glycolysis | Cytoplasm | Glucose broken down into 2 pyruvates | 2 NADH | 2 ATP |
Acetyl CoA formation | Mitochondria Matrix | Pyruvate combined with CoA to form acetyl CoA and CO2 | 2 NADH | |
Krebs Cycle | Mitochondria Matrix | Acetyl group of acetyl CoA metabolized to 2 CO2 | 6 NADH, 2 FADH2 | 2 ATP |

Additional Pathways: Fatty Acid and Amino Acid Catabolism
Fatty Acid β-Oxidation
Fatty acids are transported into the mitochondria via the carnitine shuttle and undergo β-oxidation, producing acetyl CoA, NADH, and FADH2 for entry into the citric acid cycle and electron transport chain.
Each cycle: Removes two carbons as acetyl CoA, generating one NADH and one FADH2.
Protein Catabolism
Proteins are degraded by proteases and peptidases into amino acids, which can be deaminated and converted into intermediates that enter the citric acid cycle.
Enzymes: Endopeptidases and exopeptidases
Key Equations
The following equations summarize the energetics of electron transfer from NADH and FADH2 to oxygen:
Conclusion
Aerobic respiration is a highly efficient process for extracting energy from organic molecules. The integration of glycolysis, pyruvate oxidation, the citric acid cycle, and oxidative phosphorylation allows cells to maximize ATP yield, supporting the energy demands of eukaryotic life.
