 Back
BackBioenergetics: The Flow of Energy in the Cell
Study Guide - Smart Notes

Bioenergetics: The Flow of Energy in the Cell
Introduction to Cellular Energy
Cells require energy to perform a variety of essential biological functions. The study of bioenergetics explores how energy flows through cells and the biosphere, enabling life processes. Energy is necessary for six major kinds of biological work, each critical for cellular and organismal function.
Six Kinds of Biological Work
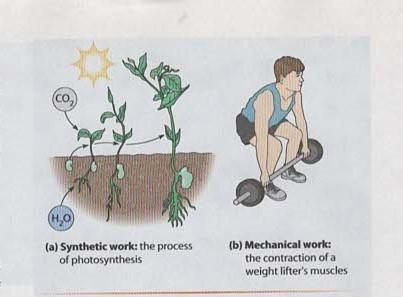
Synthetic Work: The synthesis of new molecules, such as the process of photosynthesis in plants, where solar energy is used to convert carbon dioxide and water into glucose.
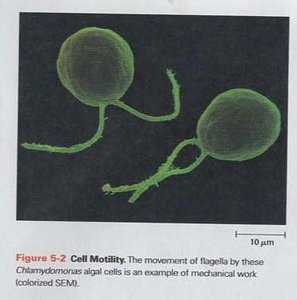
Mechanical Work: The physical movement of cellular structures or whole organisms, such as muscle contraction or the movement of flagella in cells.
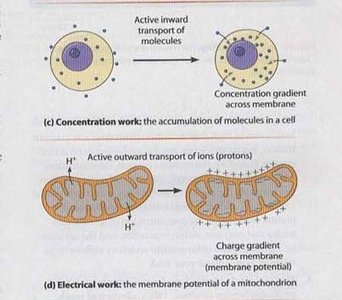
Concentration Work: The active transport of molecules across membranes to create concentration gradients, essential for nutrient uptake and waste removal.
Electrical Work: The generation and maintenance of membrane potentials, such as the charge gradient across mitochondria.
Heat Production: The generation of heat, for example, shivering in cold environments to maintain body temperature.
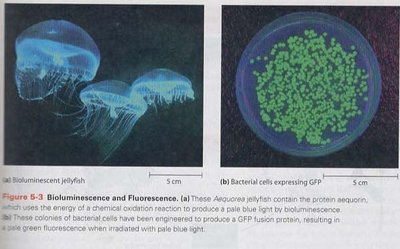
Bioluminescence: The production of light by living organisms, such as fireflies or jellyfish, often used for communication or predation.

Energy Flow Through the Biosphere
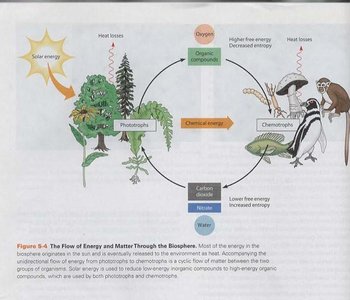
Energy flows continuously through the biosphere, originating from the sun and transferred through various biological processes. Phototrophs (such as plants) capture solar energy and convert it into chemical energy, while chemotrophs (such as animals) utilize organic compounds for energy. The flow of energy is accompanied by the cycling of matter, including carbon dioxide, water, and organic compounds.
Oxidation: The removal of electrons from a substance, often accompanied by the addition of oxygen. For example, glucose oxidation to carbon dioxide:
Reduction: The addition of electrons to a substance, often accompanied by the addition of hydrogen. For example, carbon dioxide reduction to glucose:
Systems, Heat, and Work
Understanding energy flow requires knowledge of systems, heat, and work. A system is the portion of the universe under study, while the surroundings are everything else. Systems can be:
Open: Exchange energy with surroundings (e.g., living organisms).
Closed: Do not exchange energy with surroundings.
Work is the use of energy to drive processes other than heat flow, such as mechanical movement or chemical synthesis.

The First Law of Thermodynamics: Conservation of Energy
The First Law of Thermodynamics states that energy is conserved in every physical or chemical change. The total energy in the universe remains constant, though its form may change. For a system:
Internal Energy (E): Total energy stored within a system.
Change in Internal Energy: or
Enthalpy (H): Heat content, (where P is pressure and V is volume).
Change in Enthalpy:
If is negative, the reaction is exothermic (releases heat). If is positive, the reaction is endothermic (absorbs heat).
The Second Law of Thermodynamics: Directionality and Entropy
The Second Law of Thermodynamics states that the universe tends toward greater disorder or randomness, known as entropy (S). Thermodynamic spontaneity measures whether a reaction can occur, but not whether it will occur.
Entropy (S): Measure of disorder; is the change in disorder.
Free Energy (G): Energy available to do work at constant volume and pressure;
Relationship: (where T is temperature in Kelvin)
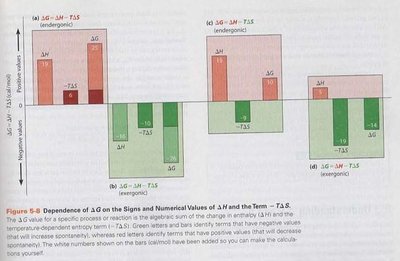
Free Energy Change and Thermodynamic Spontaneity
Spontaneous reactions are characterized by a decrease in free energy () and an increase in entropy (). Reactions can be classified as:
Exergonic: Energy-yielding, is negative.
Endergonic: Energy-requiring, is positive.
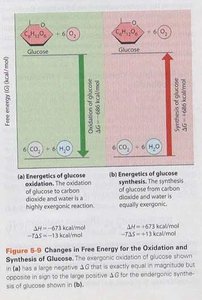
Biological Examples of Free Energy Change
Glucose oxidation is a highly exergonic reaction, releasing energy as glucose is converted to carbon dioxide and water. Conversely, the synthesis of glucose from carbon dioxide and water is endergonic, requiring energy input.
Reaction | ΔG (kcal/mol) | ΔH (kcal/mol) | -TΔS (kcal/mol) |
|---|---|---|---|
Glucose Oxidation | -686 | -673 | -13 |
Glucose Synthesis | +686 | +673 | +13 |
Summary Table: Types of Biological Work
Type of Work | Description | Example |
|---|---|---|
Synthetic | Molecule synthesis | Photosynthesis |
Mechanical | Movement | Muscle contraction, flagella movement |
Concentration | Transport across membranes | Active transport |
Electrical | Membrane potential | Mitochondrial charge gradient |
Heat Production | Thermogenesis | Shivering |
Bioluminescence | Light production | Fireflies, jellyfish |
Additional info: Academic context was added to clarify definitions, examples, and equations, and to ensure completeness for exam preparation.
