 Back
BackBioenergetics: The Flow of Energy in the Cell
Study Guide - Smart Notes

Bioenergetics: The Flow of Energy in the Cell
Understanding ΔG and Keq
Bioenergetics is the study of the transformation and flow of energy in biological systems. Two fundamental concepts in bioenergetics are the equilibrium constant (Keq) and the free energy change (ΔG), which together determine the directionality and spontaneity of cellular reactions.
Equilibrium Constant (Keq): Keq is the ratio of product concentration to reactant concentration at equilibrium. For a reaction A → B, Keq = [B]eq / [A]eq.
Directionality: If the ratio of products to reactants differs from Keq, the reaction will move toward equilibrium.
Example: For the interconversion of glucose-6-phosphate (G6P) and fructose-6-phosphate (F6P), Keq = 0.5.
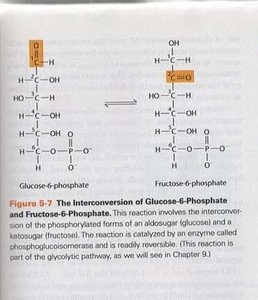
Additional info: The reaction is catalyzed by phosphoglucose isomerase and is part of glycolysis.
Free Energy and Chemical Equilibrium
The free energy of a system is lowest at equilibrium and increases as the system is displaced from equilibrium. The tendency toward equilibrium provides the driving force for all chemical reactions.
ΔG (Free Energy Change): Indicates whether a reaction is spontaneous. Negative ΔG means the reaction is thermodynamically favorable.
Driving Force: Reactions proceed spontaneously toward equilibrium, releasing energy.
Calculating ΔG
ΔG can be calculated for reactions with one or multiple reactants and products. The equations below show how ΔG relates to Keq and the concentrations of reactants and products.
For A → B:
For aA + bB → cC + dD:
Example Calculation: For G6P → F6P, Keq = 0.5, [G6P] = 10µM, [F6P] = 1µM:
Interpretation: The reaction proceeds spontaneously and releases 954 calories as long as the initial concentrations are maintained.
Standard Free Energy Change (ΔG°')
ΔG°' is the free energy change measured under standard conditions: 25°C (298K), 1 atmosphere pressure, and all reactants/products at 1M concentration. The prime (') indicates standard hydrogen ion concentration (pH 7).
At 25°C, RT = 592 cal/mol, so
ΔG°' is useful for standardized reporting but does not reflect actual cellular conditions.
ΔG' is used to measure how far from equilibrium a reaction is under real cellular concentrations.
The Relationship Between ΔG°' and K'eq
ΔG°' is directly related to the equilibrium constant. ΔG' incorporates actual concentrations, providing a more accurate measure of spontaneity in the cell.
Example: In red blood cells, [G6P] = 83µM, [F6P] = 14µM:
Interpretation: The conversion of G6P to F6P is spontaneous and releases 644 cal/mol in red blood cells.
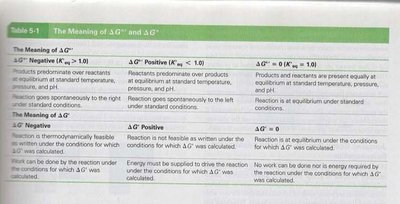
The Meaning of ΔG°' and ΔG'
ΔG°' and ΔG' provide insight into the spontaneity and directionality of reactions under standard and cellular conditions, respectively.
The Meaning of ΔG°' | ΔG°' Negative (K'eq > 1.0) | ΔG°' Positive (K'eq < 1.0) | ΔG°' = 0 (K'eq = 1.0) |
|---|---|---|---|
Products predominate over reactants at equilibrium, standard temperature, pressure, and pH. Reaction goes spontaneously to the right under standard conditions. | Reactants predominate over products at equilibrium, standard temperature, pressure, and pH. Reaction goes spontaneously to the left under standard conditions. | Products and reactants are present equally at equilibrium under standard temperature, pressure, and pH. Reaction is at equilibrium under standard conditions. | |
The Meaning of ΔG' | ΔG' Negative | ΔG' Positive | ΔG' = 0 |
Reaction is thermodynamically feasible as written under the conditions for which ΔG' was calculated. Work can be done by the reaction under the conditions for which ΔG' was calculated. | Reaction is not feasible as written under the conditions for which ΔG' was calculated. Energy must be supplied to drive the reaction under the conditions for which ΔG' was calculated. | Reaction is at equilibrium under the conditions for which ΔG' was calculated. No work can be done nor is energy required by the reaction under the conditions for which ΔG' was calculated. |
Jumping Bean Analogy for Bioenergetics
The jumping bean analogy is used to illustrate the concepts of equilibrium, enthalpy, entropy, and the capacity to do work in bioenergetics. Beans moving between compartments represent molecules transitioning between states, driven by energy changes.
Equilibrium Constant: The distribution of beans at equilibrium reflects Keq.
Enthalpy Change: The energy required for beans to move between compartments.
Entropy Change: The randomness or disorder in the distribution of beans.
ΔG and Work: The ability to do work is related to the free energy change.
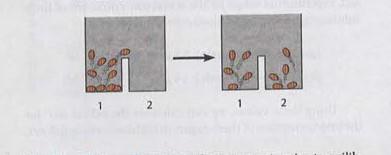
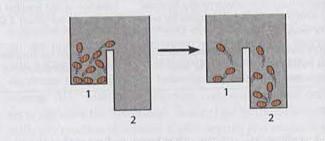
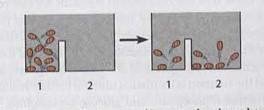
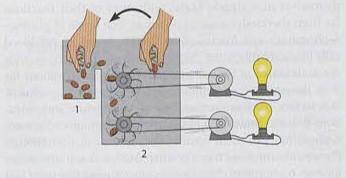
Additional info: The analogy helps visualize how energy transformations drive molecular movement and cellular processes.
