 Back
BackBioenergetics: The Flow of Energy in the Cell
Study Guide - Smart Notes

Bioenergetics: The Flow of Energy in the Cell
Introduction to Bioenergetics
Bioenergetics is the study of how energy flows through living systems, particularly cells. Understanding how cells acquire, transform, and utilize energy is fundamental to cell biology, as all cellular processes require energy input or output.
Key Concepts in Cellular Bioenergetics
Energy and Work in Biological Systems
Energy is the capacity to do work or cause specific chemical or physical changes.
Living systems require a continuous supply of energy to maintain order and drive essential processes.
Energy is required for six main categories of biological work:
Synthetic work: Formation of new chemical bonds and molecules (biosynthesis).
Mechanical work: Movement of cells or cellular components.
Concentration work: Accumulation of molecules against a concentration gradient.
Electrical work: Movement of ions to create charge gradients across membranes.
Generation of heat: Release of heat as a by-product of metabolic reactions.
Generation of light: Bioluminescence in certain organisms.
Synthetic Work
Synthetic work involves the process of biosynthesis, such as the formation of macromolecules from smaller precursors. This is essential for cell growth and maintenance.
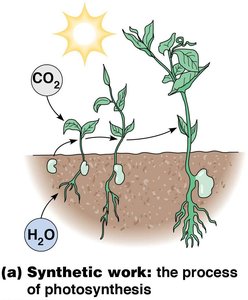
Mechanical Work
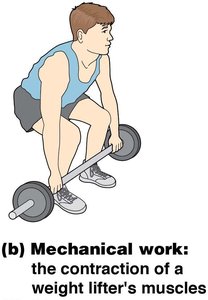
Mechanical work refers to the movement of cells or subcellular structures, such as muscle contraction, chromosome movement during mitosis, and cytoplasmic streaming.
Concentration Work
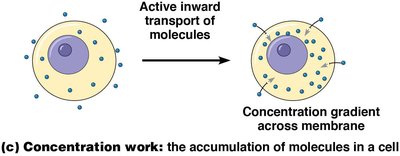
Concentration work is the active transport of molecules across membranes, creating and maintaining concentration gradients essential for cellular function.
Electrical Work
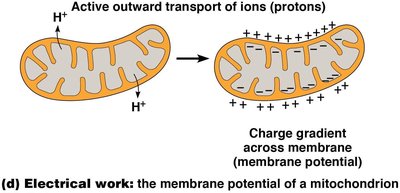
Electrical work involves the movement of ions across membranes, generating membrane potentials critical for processes like ATP synthesis and nerve impulse transmission.
Generation of Heat
Heat is produced as a by-product of metabolic reactions, especially in endothermic (warm-blooded) animals, which use metabolic heat to regulate body temperature.

Systems, Surroundings, and the Universe
In thermodynamics, the system is the part of the universe under study, while the surroundings are everything else. The system and surroundings together make up the universe. Cells are considered open systems because they exchange both matter and energy with their surroundings.
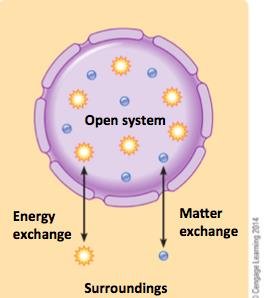
Thermodynamic Laws in Biology
The First Law of Thermodynamics
The first law states that energy cannot be created or destroyed, only transformed. The total energy of the universe remains constant. In cells, energy transformations are central to metabolism.
Internal energy (E): Total energy stored within a system.
Change in internal energy:
For chemical reactions:
Enthalpy (H)
Enthalpy is the heat content of a system at constant pressure. In biological systems, changes in enthalpy () are often equivalent to changes in internal energy.
At constant pressure:
Exothermic reactions () release heat; endothermic reactions () absorb heat.
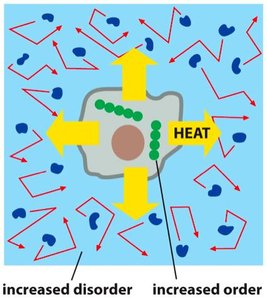
The Second Law of Thermodynamics
The second law states that the entropy (disorder) of the universe always increases in spontaneous processes. Entropy (S) is a statistical measure of disorder or randomness.
Spontaneous processes move from less probable (ordered) to more probable (disordered) states.
Maintaining order (such as concentration gradients) in cells requires continuous energy input.
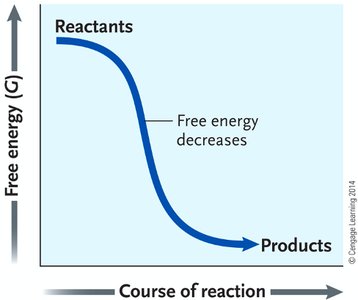
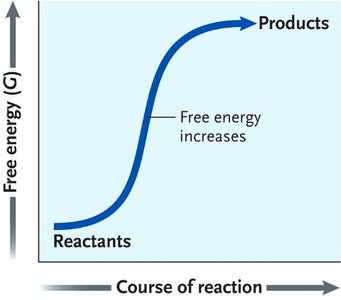
Gibbs Free Energy (G)
Gibbs free energy determines whether a process is thermodynamically spontaneous in a system. The change in free energy () is given by:
If , the process is exergonic (spontaneous); if , it is endergonic (nonspontaneous); if , the system is at equilibrium.
Equilibrium and Free Energy
Chemical Equilibrium and the Equilibrium Constant (Keq)
At equilibrium, the rates of the forward and reverse reactions are equal, and there is no net change in reactant or product concentrations. The equilibrium constant () is defined as:
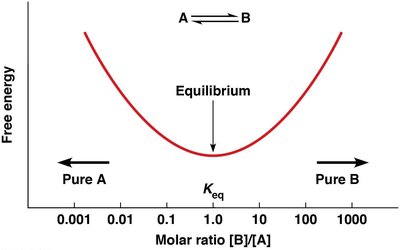
Relationship Between ΔG and Keq
The free energy change for a reaction under non-standard conditions is related to the equilibrium constant and the actual concentrations of reactants and products:
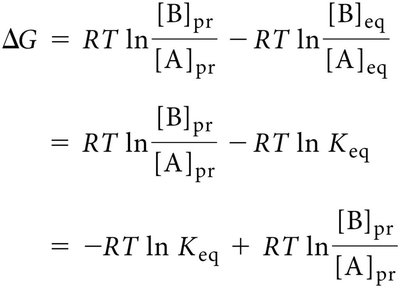
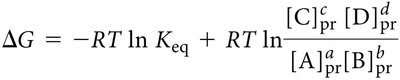
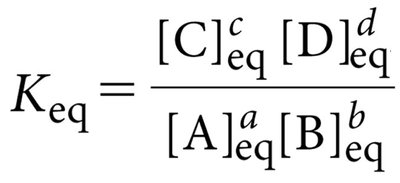
Standard Free Energy Change (ΔGº′)
ΔGº′ is the free energy change under standard conditions (298 K, 1 atm, 1 M concentrations, pH 7.0 for biological systems). It provides a reference point for comparing reactions but does not reflect actual cellular conditions.
Free Energy Change in Cells (ΔG′)
ΔG′ is the free energy change under prevailing cellular conditions, using the actual concentrations of reactants and products. This value determines whether a reaction is spontaneous in the cell.
Example: Glycolysis Step
The conversion of glucose-6-phosphate to fructose-6-phosphate (step two of glycolysis) is catalyzed by phosphoglucoisomerase. The equilibrium constant and free energy changes can be calculated as follows:
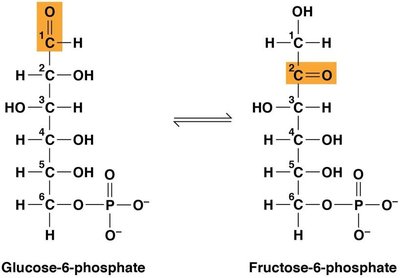
For K′eq = 0.5,
In red blood cells, the actual concentrations make ΔG′ negative, so the reaction proceeds spontaneously in vivo, even if ΔGº′ is positive.
Coupling Reactions in Cells
Coupling Exergonic and Endergonic Processes
Cells drive nonspontaneous (endergonic) processes by coupling them to spontaneous (exergonic) processes. The overall free energy change for coupled reactions is the sum of the individual ΔG values. If the total ΔG is negative, the coupled process is spontaneous.
Mechanisms of Coupling
Sequential coupling: Linking reactions in a pathway.
Physical coupling: Direct transfer of energy between molecules.
Activated carriers: Molecules like ATP that store and transfer energy.
Thermodynamics vs. Kinetics
Spontaneity and Reaction Rates
Thermodynamics determines whether a reaction can occur (spontaneity), but not how fast it will occur (rate). Kinetics, which involves activation energy and reaction pathways, determines the rate of a reaction.
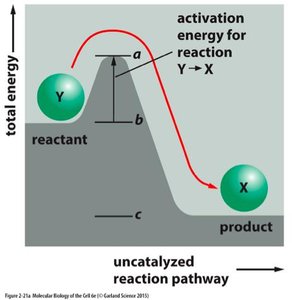
Even if ΔG < 0, a reaction may proceed slowly if the activation energy is high.
Enzymes lower activation energy, increasing reaction rates without affecting ΔG.
Summary Table: Types of Biological Work
Type of Work | Description | Example |
|---|---|---|
Synthetic | Formation of new molecules | Photosynthesis |
Mechanical | Movement of structures | Muscle contraction |
Concentration | Transport against gradients | Active transport of glucose |
Electrical | Ion movement, membrane potential | Proton pumping in mitochondria |
Heat | Release of thermal energy | Shivering |
Light | Bioluminescence | Fireflies |
Additional info: The notes above integrate foundational thermodynamic principles with their application to cellular processes, as required for a college-level cell biology course. All equations are provided in LaTeX format, and only directly relevant images are included to reinforce key concepts.
