 Back
BackBioenergetics: The Flow of Energy in the Cell (Chapter 5 Study Guide)
Study Guide - Smart Notes

Bioenergetics: The Flow of Energy in the Cell
Introduction to Bioenergetics
Bioenergetics is the study of how energy flows and is transformed within living cells. Understanding these principles is essential for grasping how cells perform work, maintain structure, and drive life processes. The laws of thermodynamics, especially the concepts of energy conservation and entropy, are foundational to bioenergetics.
Energy: The capacity to cause chemical or physical change.
Bioenergetics: Application of thermodynamic principles to biological systems.
Thermodynamics: Governs energy transactions in physical and chemical processes.
Standard Physiological Conditions
Definition and Importance
Standard physiological conditions are reference points for biochemical reactions, but actual cellular conditions often differ. These include:
Temperature: 37°C (for human cells)
pH: Approximately 7.2
Pressure: 1 atm
Water concentration: 55 M
Cells maintain conditions far from equilibrium to drive reactions in the direction needed for life.
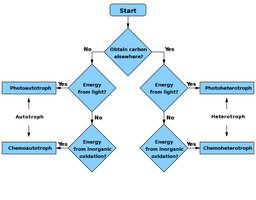
Classification of Organisms Based on Bioenergetics
Energy and Carbon Sources
Organisms are classified by how they obtain energy and carbon. This classification is crucial for understanding metabolic diversity and ecological roles.
Phototrophs: Use light energy.
Chemotrophs: Use chemical energy from organic or inorganic compounds.
Autotrophs: Use CO2 as a carbon source.
Heterotrophs: Use organic molecules as a carbon source.
Type | Energy Source | Carbon Source |
|---|---|---|
Photoautotroph | Light | CO2 |
Chemoautotroph | Inorganic oxidation | CO2 |
Photoheterotroph | Light | Organic molecules |
Chemoheterotroph | Inorganic oxidation | Organic molecules |
Metabolism: Anabolic and Catabolic Pathways
Overview of Metabolic Pathways
Metabolism encompasses all chemical reactions in a cell, divided into two main types:
Anabolism: Building up complex molecules from simpler ones; requires energy (endergonic).
Catabolism: Breaking down complex molecules into simpler ones; releases energy (exergonic).
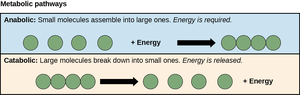
Example: Protein synthesis (anabolism) vs. glycolysis (catabolism).
Chemical Potential Energy
Definition and Examples
Chemical potential energy is the energy stored in the bonds of molecules. It is released during metabolic reactions, often as heat or to drive cellular work.
ATP: High-energy phosphate bonds.
Glucose: C-H bonds.
Fats: C-H bonds.

Application: Energy from glucose oxidation powers cellular activities.
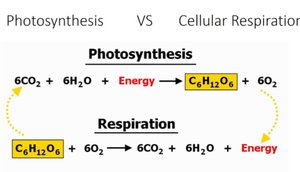
Symbiotic Relationship Between Phototrophs and Chemotrophs
Carbohydrate Metabolism
Phototrophs produce carbohydrates via photosynthesis, which chemotrophs use for energy through cellular respiration. This relationship is fundamental to the biosphere's energy flow.
Exergonic and Endergonic Reactions
Spontaneity and Energy Changes
Biochemical reactions are classified by their energy profiles:
Exergonic: Energy-releasing, spontaneous ().
Endergonic: Energy-requiring, non-spontaneous ().
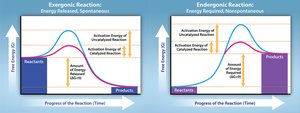
Example: ATP hydrolysis (exergonic) vs. glucose phosphorylation (endergonic).
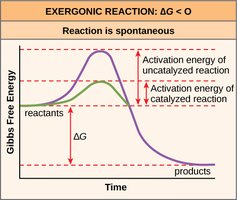
Enzyme Catalysis and Activation Energy
Role of Enzymes
Enzymes are biological catalysts that lower the activation energy required for reactions, enabling them to proceed at physiologically relevant rates.
Activation Energy: The energy barrier that must be overcome for a reaction to occur.
Enzyme Action: Stabilizes the transition state, increases reaction speed.
Potential and Kinetic Energy in Biological Systems
Definitions and Examples
Cells utilize both potential and kinetic energy:
Potential Energy: Stored energy (e.g., in chemical bonds).
Kinetic Energy: Energy of motion (e.g., muscle contraction).

Example: Muscle fibers use ATP (potential energy) to contract (kinetic energy).
Types of Cellular Work
Six Categories of Cellular Work
Cells require energy to perform various types of work:
Synthetic Work: Formation of new chemical bonds (biosynthesis).
Mechanical Work: Movement of cells or subcellular structures.
Concentration Work: Moving molecules against concentration gradients.
Electrical Work: Moving ions against electrochemical gradients.
Generation of Heat: Maintaining body temperature in homeotherms.
Generation of Light: Bioluminescence and fluorescence.
Bioluminescence and Fluorescence in Cell Biology
Green Fluorescent Protein (GFP)
Bioluminescence is the production of light by living organisms, often through ATP or chemical oxidation. GFP, derived from jellyfish, is widely used in cell biology to study protein localization and function.
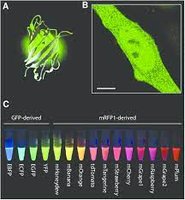
Application: GFP fusion proteins allow visualization of cellular processes.
Energy Flow and Matter Cycling in the Biosphere
Photosynthesis and Cellular Respiration
Energy flows unidirectionally through the biosphere, beginning with photosynthesis and ending with cellular respiration. Matter cycles between phototrophs and chemotrophs.

Example: Plants synthesize glucose (photosynthesis), animals oxidize glucose (respiration).
Thermodynamic Principles in Biological Systems
First and Second Laws of Thermodynamics
Biological systems obey the laws of thermodynamics:
First Law: Energy is conserved; it cannot be created or destroyed.
Second Law: Entropy (disorder) increases in isolated systems; reactions have directionality.
Entropy (S): Measure of disorder; spontaneous reactions increase entropy.
Gibbs Free Energy and Reaction Spontaneity
ΔG, ΔH, and ΔS
Gibbs free energy () determines whether a reaction is spontaneous:
ΔH: Enthalpy change (heat content)
ΔS: Entropy change (disorder)
T: Temperature in Kelvin
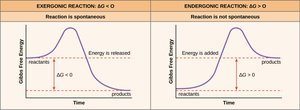
Example: Glucose oxidation () is spontaneous; photosynthesis () is not.
Coupled Reactions in Cellular Metabolism
Mechanism and Importance
Cells often couple exergonic and endergonic reactions to drive essential processes. ATP hydrolysis is a common exergonic reaction used to power endergonic steps, such as glucose phosphorylation in glycolysis.
ATP Hydrolysis:
Glucose Phosphorylation: Endergonic, driven by ATP hydrolysis
Example: Sodium-potassium pump, muscle contraction, biosynthesis.
Practice Questions and Applications
Sample Quiz Questions
What type of energy is stored in ATP? Potential energy
What is the primary role of ATP in cells? Energy currency for cellular processes
Which process is an example of an endergonic reaction? Photosynthesis
How is glucose phosphorylation made favorable? By coupling with ATP hydrolysis
Summary
Bioenergetics is central to cell biology, explaining how cells manage energy to perform work, maintain order, and drive life processes. Key concepts include metabolic pathways, energy flow, thermodynamic principles, and the importance of coupled reactions in cellular metabolism.
