 Back
BackCell Signaling Pathways: Electrical and Chemical Signaling in Neurons
Study Guide - Smart Notes

Signaling Pathways in Neurons
Overview of Nerve Cell Signaling
Neurons communicate using both electrical and chemical signals, allowing rapid and regulated information transfer throughout the nervous system. Key mechanisms include voltage changes across membranes, ion channel activity, and receptor-mediated signaling.
Electrical signaling involves changes in membrane potential due to ion movement.
Chemical signaling involves neurotransmitter release and receptor activation.
Major receptor types: GPCRs (G protein-coupled receptors), RTKs (receptor tyrosine kinases), and ligand-gated ion channels.
Structure and Function of Neurons
Basic Anatomy of a Motor Neuron
Neurons are specialized cells with distinct regions for receiving, integrating, and transmitting signals.
Dendrites: Receive incoming signals.
Cell body (soma): Contains the nucleus and integrates signals.
Axon: Conducts electrical impulses away from the cell body.
Axon hillock: Site where action potentials are initiated.
Terminal branches and bulbs: Transmit signals to other neurons, muscles, or glands.
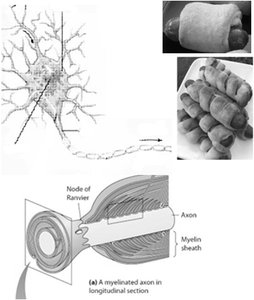
Myelin sheath: Insulates axons to speed up signal transmission.
Synapse: Junction between two neurons for signal transmission.
Membrane Potential and Ion Distribution
Resting Membrane Potential
The resting membrane potential (Vm) is the electrical potential difference across the plasma membrane of a resting cell, typically negative inside relative to outside.
Caused by unequal distribution of ions and selective membrane permeability.
Typical value: -60 mV (e.g., squid giant axon).
Major contributors: trapped negatively charged macromolecules, Na+/K+ pump, and K+ permeability.

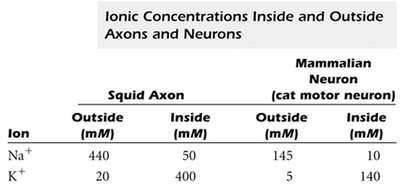
Ionic Concentrations Inside and Outside Neurons
Ion gradients are essential for generating membrane potentials and action potentials.
Ion | Squid Axon Outside (mM) | Squid Axon Inside (mM) | Mammalian Neuron Outside (mM) | Mammalian Neuron Inside (mM) |
|---|---|---|---|---|
Na+ | 440 | 50 | 145 | 10 |
K+ | 20 | 400 | 5 | 140 |
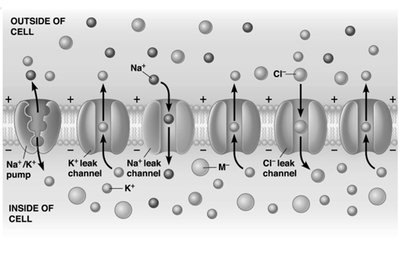
Ion Channels and Pumps
Membrane proteins regulate ion movement, maintaining gradients and enabling signaling.
Na+/K+ pump: Actively transports 3 Na+ out and 2 K+ in, using ATP.
Leak channels: Allow passive movement of K+, Na+, and Cl-.
Membrane is more permeable to K+ than Na+.
Ion Distribution and Membrane Potential
Different ions have distinct concentration gradients across the membrane.
K+: High inside, low outside
Na+: Low inside, high outside
Cl-: Low inside, high outside

Electrical Excitability and Action Potentials
Voltage-Gated Ion Channels
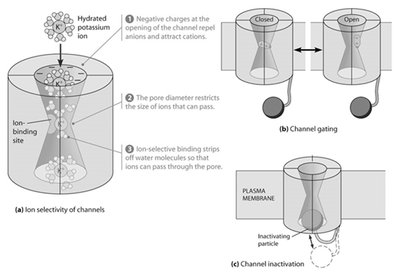
Voltage-gated channels open or close in response to changes in membrane potential, enabling rapid signaling.
Ion selectivity: Channels allow only specific ions to pass.
Channel gating: Channels switch between open and closed states.
Inactivation: Some channels become temporarily non-conductive after opening.
Action Potential Phases
An action potential is a rapid, transient change in membrane potential that propagates along the axon.
Resting phase: Vm is stable and negative.
Depolarizing phase: Na+ channels open, Na+ influx causes rapid depolarization.
Repolarizing phase: Na+ channels inactivate, K+ channels open, K+ efflux restores negativity.
Hyperpolarizing phase (undershoot): K+ efflux continues, Vm becomes more negative than resting.
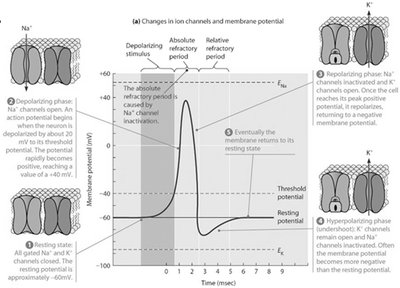
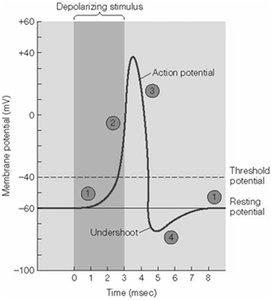
Propagation of Action Potentials
Action potentials travel unidirectionally along the axon due to sequential depolarization and repolarization.
Depolarization at one region triggers depolarization in the next.
Refractory periods prevent backward propagation.

Saltatory Conduction
In myelinated axons, action potentials jump between nodes of Ranvier, increasing conduction speed.
Myelin sheath: Insulates axon, preventing ion leakage.
Nodes of Ranvier: Gaps in myelin where action potentials are regenerated.
Synaptic Transmission
Electrical Synapses
Electrical synapses use gap junctions to allow direct ion flow between neurons, enabling rapid signal transmission.
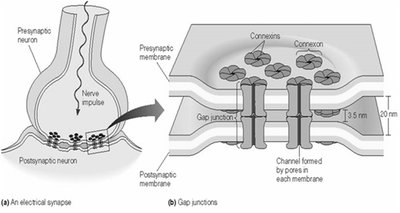
Chemical Synapses
Chemical synapses use neurotransmitters to transmit signals across a synaptic cleft.
Action potential triggers neurotransmitter release from presynaptic neuron.
Neurotransmitter binds to receptors on postsynaptic neuron, causing depolarization or hyperpolarization.
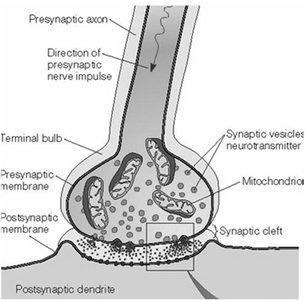
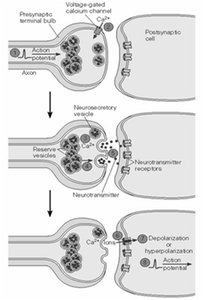
Neurotransmitters and Receptors
Neurotransmitter Function
Neurotransmitters are chemicals that transmit signals across synapses. They can be excitatory (depolarizing) or inhibitory (hyperpolarizing).
Acetylcholine: Common excitatory neurotransmitter in vertebrates; increases Na+ permeability in skeletal muscle.
Ligand-gated channels: Open in response to neurotransmitter binding (e.g., acetylcholine receptor).
Signal Integration at the Axon Hillock
The axon hillock integrates excitatory and inhibitory postsynaptic potentials (EPSPs and IPSPs) to determine if an action potential will be generated.
EPSPs: Depolarizing signals that bring membrane potential closer to threshold.
IPSPs: Hyperpolarizing signals that move membrane potential away from threshold.
Termination of Synaptic Transmission
Neurotransmitter removal resets the synapse for future signaling.
Degradation (e.g., acetylcholinesterase breaks down acetylcholine).
Reuptake into presynaptic neuron.
Signal Transduction Pathways
Ligand-Receptor Interactions
Signal transduction begins with the specific, non-covalent binding of a ligand to its receptor, initiating intracellular signaling cascades.
Affinity: Strength of ligand-receptor binding, measured by dissociation constant (Kd).
Down-regulation: Decrease in receptor number or function.
Agonists: Activate receptors; antagonists: block receptor function.
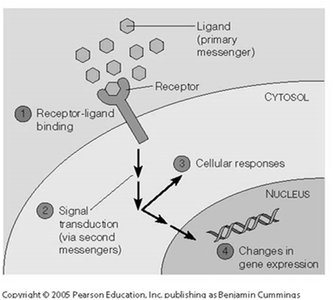
Types of Receptors
Ligand-gated channels: Open in response to ligand binding (e.g., acetylcholine receptor).
GPCRs: Activate G proteins to trigger second messenger cascades.
Receptor kinases (RTKs): Activate kinase cascades via phosphorylation.
Intracellular receptors: Bind small or hydrophobic ligands (e.g., steroid hormones).
G Protein-Coupled Receptor (GPCR) Signaling
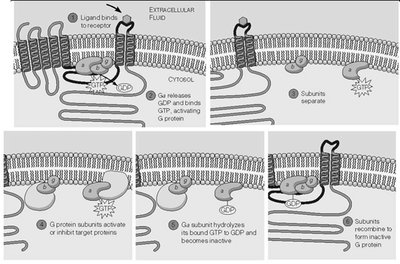
Structure and Activation of GPCRs
GPCRs are seven-transmembrane domain receptors that activate heterotrimeric G proteins upon ligand binding.
G proteins consist of α, β, and γ subunits.
GDP-bound α is inactive; GTP-bound α is active and dissociates from βγ.
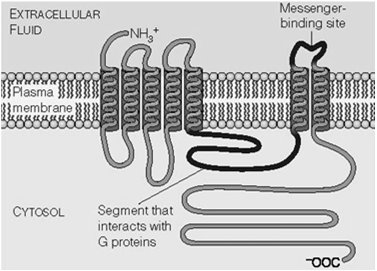
Second Messengers in GPCR Pathways
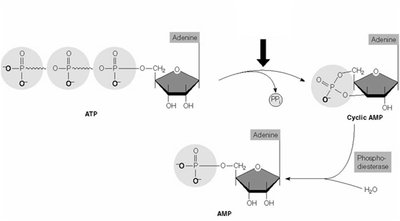
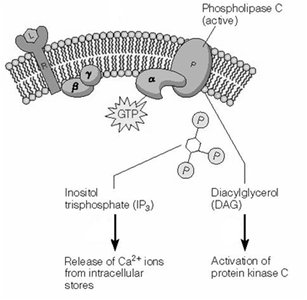
GPCR activation leads to the production of second messengers such as cAMP, IP3, DAG, and Ca2+.
cAMP: Produced by adenylyl cyclase; activates protein kinase A (PKA).
IP3 and DAG: Produced by phospholipase C; IP3 releases Ca2+ from ER, DAG activates PKC.
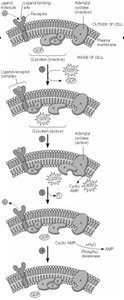
Amplification and Hierarchy in Signal Transduction
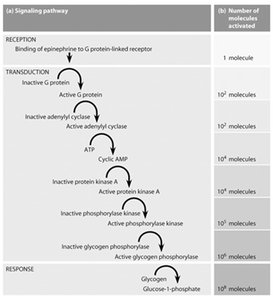
Signal transduction pathways amplify signals through cascades, allowing a small number of ligand-receptor interactions to produce large cellular responses.
Example: Epinephrine binding to GPCR leads to cAMP production, PKA activation, and glycogen breakdown in liver.
Phospholipase C Pathway and Calcium Signaling
IP3 and DAG Production
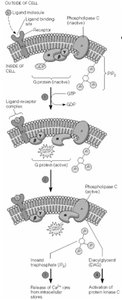
Ligand binding to GPCR can activate phospholipase C, generating IP3 and DAG from membrane phospholipids.
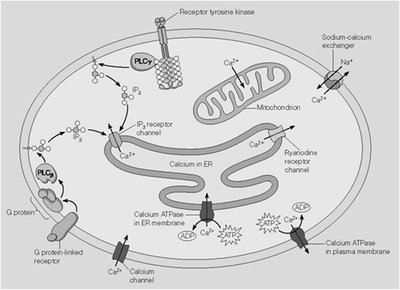
IP3 releases Ca2+ from ER by opening IP3 receptor channels.
DAG activates protein kinase C (PKC).
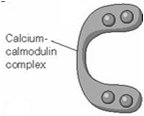
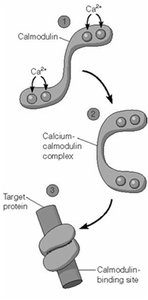
Calcium Regulation and Calmodulin
Calcium acts as a versatile second messenger, with its effects mediated by binding to proteins such as calmodulin.
At low cytosolic Ca2+ (10-4 mM), calmodulin is inactive.
At elevated Ca2+ (10-3 mM), calmodulin binds Ca2+ and activates target proteins (e.g., myosin light-chain kinase).
Receptor Tyrosine Kinases (RTKs) and Ras Pathway
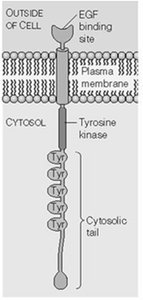
RTK Activation and Signal Transduction
RTKs are activated by ligand-induced dimerization and autophosphorylation, leading to recruitment of SH2-domain proteins and activation of downstream pathways.
Example: EGF receptor dimerizes and autophosphorylates upon ligand binding.
Phosphorylated RTKs recruit adaptor proteins (e.g., GRB2) and activate Ras.
Ras-MAPK Pathway
Activated Ras (a small G protein) initiates a kinase cascade (Raf → MEK → MAPK), ultimately leading to changes in gene expression and cell division.
GEF (Sos) activates Ras by promoting GTP binding.
MAPK phosphorylates transcription factors (e.g., Jun in AP-1 complex).
Additional info: This guide covers key aspects of neuronal signaling, including membrane potentials, action potentials, synaptic transmission, and major cell signaling pathways relevant to cell biology.
