 Back
BackCellular Energetics: Chemiosmosis, Glycolysis, Mitochondria, Electron Transport, and Photosynthesis
Study Guide - Smart Notes

Cellular Energetics
Overview of Cellular Energetics
Cellular energetics encompasses the processes by which cells convert energy from various sources into usable forms, primarily ATP. This chapter focuses on the mechanisms of energy conversion in both mitochondria and chloroplasts, including glycolysis, the citric acid cycle, electron transport, chemiosmosis, and photosynthesis.
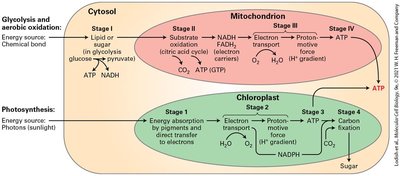
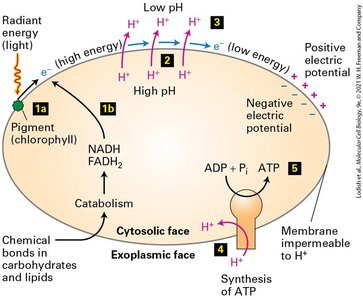
Chemiosmosis, Electron Transport, Proton-Motive Force, and ATP Synthesis
Chemiosmosis is the process by which cells convert chemical bond energy, chemical gradients, and electrical gradients across membranes into ATP. The proton-motive force is the energy stored in the proton electrochemical gradient, which is harnessed for ATP synthesis.
Chemiosmosis: Interconversion of energy forms across membranes.
Proton-motive force: Generated by electron transport chain, used by ATP synthase.
ATP synthesis: Occurs as protons flow through ATP synthase, rotating its subunits.
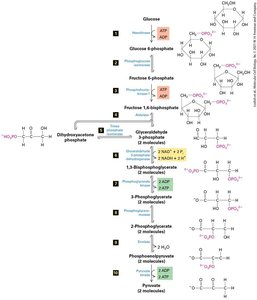
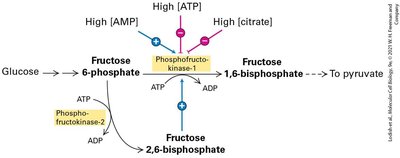
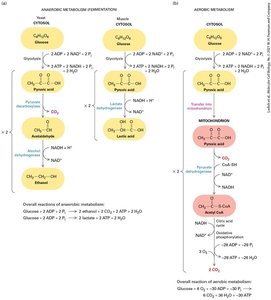
Glycolysis: First Step of Harvesting Energy from Glucose
Glycolysis is the initial stage of glucose metabolism, occurring in the cytosol. It converts glucose into two molecules of pyruvate, generating ATP and NADH. Under anaerobic conditions, pyruvate is further metabolized to lactic acid or ethanol, regenerating NAD+ for continued glycolysis.
Aerobic oxidation: Four-stage process converting glucose/fatty acids to ATP.
Glycolysis: Produces 2 ATP and 2 NADH per glucose.
Anaerobic metabolism: Pyruvate converted to lactic acid or ethanol.
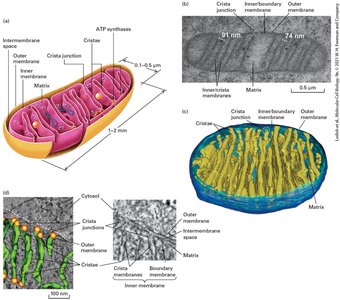
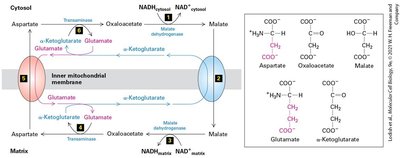
The Structure of Mitochondria
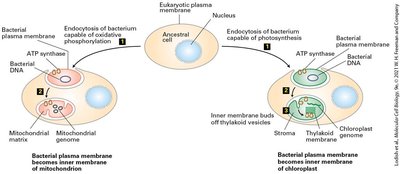
Mitochondria are double-membraned organelles central to aerobic energy production. The endosymbiont hypothesis suggests mitochondria evolved from bacteria. Mitochondria contain distinct compartments and are sites of ATP generation via aerobic oxidation.
Outer and inner membranes: Create intermembrane space and matrix.
Compartmentalization: Enables specialized metabolic functions.
Endosymbiont hypothesis: Origin of mitochondria and chloroplasts from ancestral bacteria.
Multiple Functions of Mitochondria
Mitochondria perform diverse functions beyond ATP synthesis, including biosynthesis, ion homeostasis, cell death regulation, and contributions to immunity and inflammation.
Biosynthesis/Processing | Other Functions |
|---|---|
Fatty acids, steroid hormones, pyrimidines, iron-sulfur clusters, heme, phospholipids, ubiquinone, amino acids | Oxidative phosphorylation, ROS homeostasis, ion homeostasis, ammonia detoxification, fatty acid oxidation, thermogenesis, innate immunity, apoptosis, mitochondrial dynamics, mitophagy, calcium transport, lipid import, stress responses, neurodegenerative pathology |
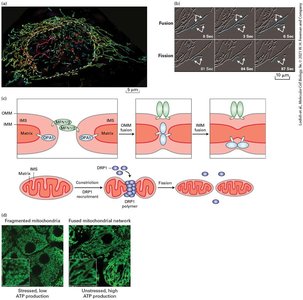
The Dynamics and Functions of Mitochondria
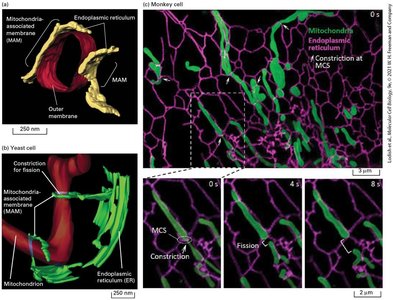
Mitochondria undergo fusion and fission, regulated by cellular state. Defective mitochondria are removed by mitophagy. Membrane contact sites (MCSs) facilitate organelle interactions, crucial for calcium and energy metabolism.
Fusion and fission: Maintain mitochondrial function and quality.
Mitophagy: Selective degradation of damaged mitochondria.
MCSs: Tethering regions for organelle communication.
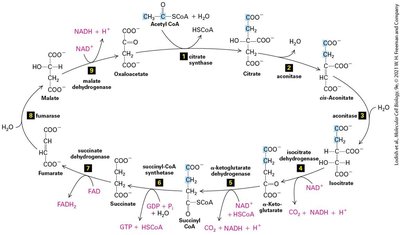
The Citric Acid Cycle and Fatty Acid Oxidation
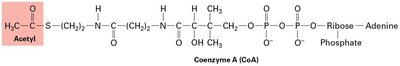
The citric acid cycle (Krebs cycle) oxidizes acetyl CoA to CO2, generating NADH and FADH2 for electron transport. Fatty acid oxidation occurs in mitochondria (short/long chains) and peroxisomes (very long chains), producing ATP or heat.
Citric acid cycle: Central metabolic pathway for energy extraction.
Fatty acid oxidation: Mitochondrial and peroxisomal pathways.
Energy carriers: NADH and FADH2 drive electron transport.
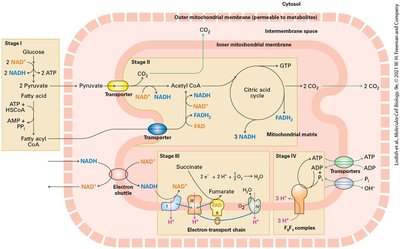
The Electron-Transport Chain and Generation of the Proton-Motive Force
Electrons from NADH and FADH2 flow through the electron transport chain, driving proton transport across the inner mitochondrial membrane and generating a proton-motive force. Reduction potentials favor unidirectional electron flow to O2.
Electron transport chain: Series of protein complexes transferring electrons.
Proton-motive force: Voltage and pH gradients across membrane.
Reduction potentials: Ensure "downhill" electron flow.
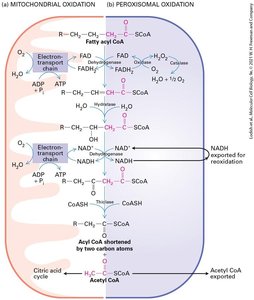
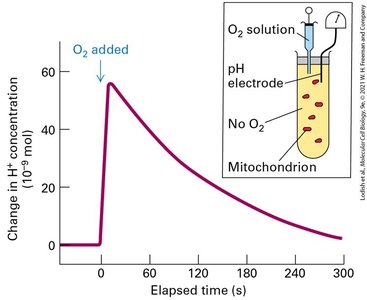
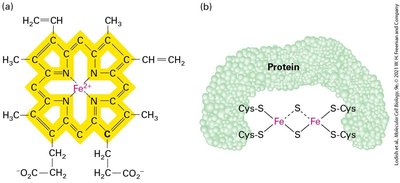
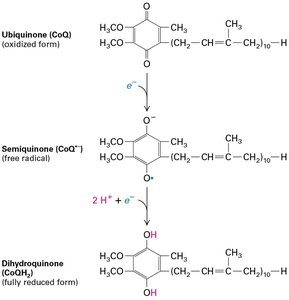
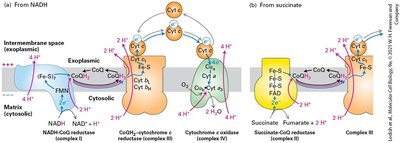
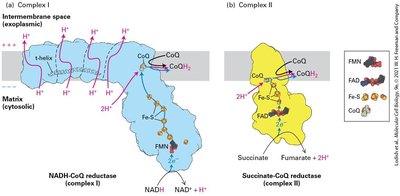
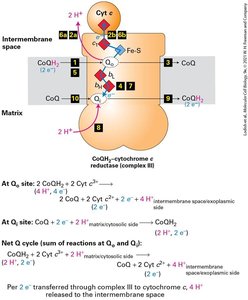
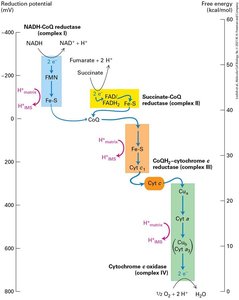
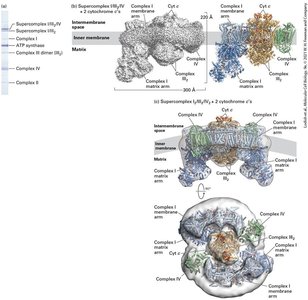
Table: Net Result of Glycolytic Pathway and Citric Acid Cycle
Reaction | CO2 Produced | NADH Produced | FADH2 Produced | ATP/GTP Produced |
|---|---|---|---|---|
Glucose to 2 pyruvate | 0 | 2 | 0 | 2 |
2 pyruvate to 2 acetyl CoA | 2 | 2 | 0 | 0 |
2 acetyl CoA to 4 CO2 | 4 | 6 | 2 | 2 |
Total | 6 | 10 | 2 | 4 |
Table: Electron-Carrying Prosthetic Groups in the Electron-Transport Chain
Protein Component | Prosthetic Groups |
|---|---|
NADH-CoQ reductase (Complex I) | FMN, Fe-S |
Succinate-CoQ reductase (Complex II) | FAD, Fe-S |
CoQH2-cytochrome c reductase (Complex III) | Heme bL, Heme bH, Fe-S, Heme C1 |
Cytochrome c | Heme c |
Cytochrome c oxidase (Complex IV) | Cua2+, Heme a, Cub2+, Heme a3 |
Harnessing the Proton-Motive Force to Synthesize ATP
The chemiosmotic hypothesis states that the proton-motive force across the inner mitochondrial membrane is the immediate energy source for ATP synthesis. ATP synthase (F0F1 complex) catalyzes ATP formation as protons flow through and rotate its subunits.
ATP synthase: Universal enzyme in bacteria, mitochondria, and chloroplasts.
Proton flow: Drives rotation and ATP synthesis.
Chloroplasts and Photosynthesis
Photosynthesis in plants produces O2 and carbohydrates. Light-capturing and ATP-generating reactions occur in thylakoid membranes. The process includes four stages: light absorption, electron transport, ATP synthesis, and carbon fixation.
Principal products: O2, starch, sucrose.
Stages: Light absorption, electron transport, ATP synthesis, carbon fixation.
Use of Light Energy to Generate Molecular Oxygen, NADPH, and ATP
Photosystems PSI and PSII in chloroplasts have distinct functions: PSII generates O2 from H2O, PSI reduces NADP+ to NADPH. Light energy is transferred to chlorophyll a in reaction centers, driving cyclic electron flow and proton-motive force for ATP synthesis.
ATP and NADPH Drive Carbon Fixation in the Calvin Cycle
The Calvin cycle fixes CO2 into organic molecules in the chloroplast stroma. C3 plants lose CO2 during photorespiration, while C4 plants minimize this loss by shuttling four-carbon molecules to bundle sheath cells for the Calvin cycle.
Calvin cycle: Series of reactions for carbon fixation.
Photorespiration: Loss of CO2 in C3 plants.
C4 plants: Efficient CO2 fixation, reduced photorespiration.
Key Equations
ATP synthesis:
Glycolysis net reaction:
Citric acid cycle net reaction:
Photosynthesis overall:
Additional info: Academic context was added to clarify the roles of mitochondria, the electron transport chain, and photosynthesis, and to ensure completeness for exam preparation.
