 Back
BackChemotrophic Energy Metabolism: Glycolysis and Fermentation
Study Guide - Smart Notes

Chemotrophic Energy Metabolism: Glycolysis and Fermentation
Overview of Cellular Energy Metabolism
Chemotrophic energy metabolism refers to the processes by which cells extract energy from chemical compounds, primarily through glycolysis and fermentation. These pathways are central to cellular bioenergetics, enabling cells to convert glucose and other substrates into usable energy in the form of ATP.
ATP: The Universal Energy Coupler
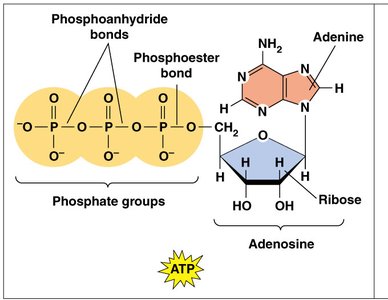
ATP (adenosine triphosphate) is the primary energy currency of the cell. It stores energy in its high-energy phosphate bonds, which can be hydrolyzed to release energy for cellular work.
Structure: ATP consists of adenosine (adenine + ribose) and three phosphate groups linked by phosphoanhydride bonds.
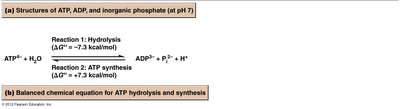
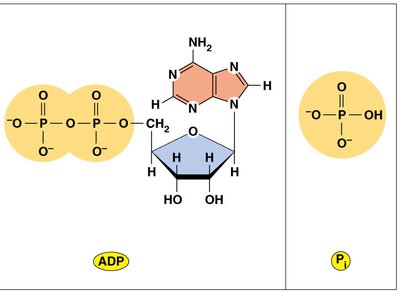
Hydrolysis: The hydrolysis of ATP to ADP and inorganic phosphate (Pi) releases energy ( kcal/mol).
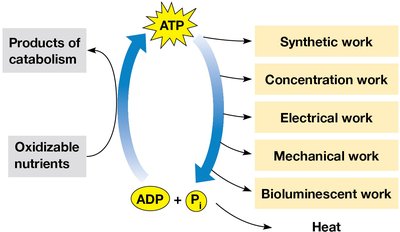
Function: ATP is used for synthetic, mechanical, electrical, and concentration work in cells.

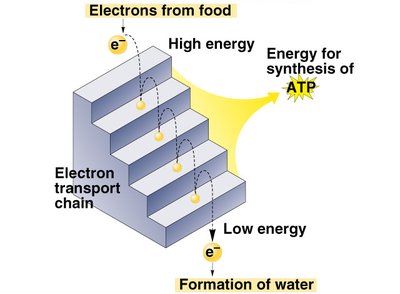
Redox Reactions in Cellular Metabolism
Redox reactions are fundamental to energy metabolism. Oxidation involves the loss of electrons, while reduction involves the gain of electrons. These reactions drive the transfer of energy within the cell.
NAD+: Nicotinamide adenine dinucleotide acts as an electron carrier, accepting electrons during glycolysis and fermentation.
Glucose Oxidation: The complete oxidation of glucose is highly exergonic ( kcal/mol).
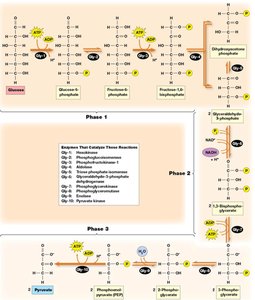
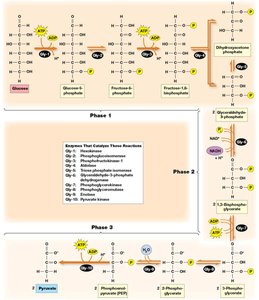
Glycolysis: Processing Glucose to Pyruvate
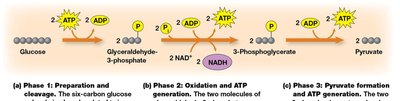
Glycolysis is a ten-step pathway occurring in the cytoplasm, converting glucose into pyruvate. It can function under both aerobic and anaerobic conditions and is divided into three phases.
Net Yield: For each glucose molecule, glycolysis produces 2 pyruvate, 2 ATP, and 2 NADH.
Phases: Preparation and cleavage, oxidation and ATP generation, pyruvate formation and ATP generation.
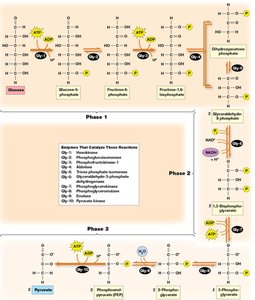
Key Glycolytic Reactions
Reaction 1: Phosphorylation of glucose to glucose-6-phosphate by hexokinase (irreversible, traps glucose in cell).
Reaction 3: Phosphorylation of fructose-6-phosphate to fructose-1,6-bisphosphate by phosphofructokinase (rate-limiting, irreversible).
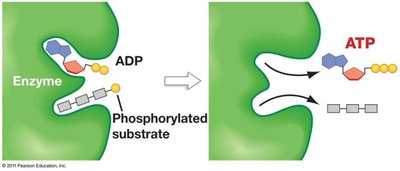
Substrate-Level Phosphorylation
ATP is generated directly in glycolysis by substrate-level phosphorylation, where a phosphate group is transferred from a high-energy substrate to ADP.
Summary Equation for Glycolysis
The overall reaction for glycolysis is:

Catabolic Fates of Pyruvate
Pyruvate produced by glycolysis can be further metabolized depending on the presence or absence of oxygen.
Aerobic Respiration: Pyruvate is converted to acetyl-CoA and enters the citric acid cycle.
Anaerobic Respiration: Pyruvate undergoes fermentation to regenerate NAD+.

Fermentation Pathways
Fermentation allows cells to regenerate NAD+ in the absence of oxygen, enabling glycolysis to continue.
Alcohol Fermentation: Occurs in yeast, converting pyruvate to ethanol and CO2.
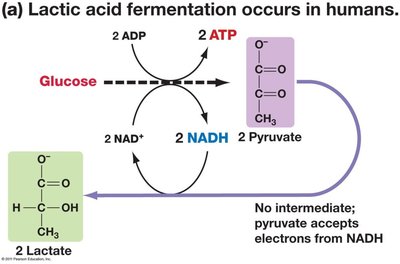
Lactic Acid Fermentation: Occurs in humans, converting pyruvate to lactate.
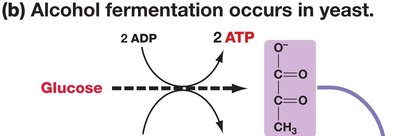

Summary Equations for Fermentation
Alcohol Fermentation:
Lactic Acid Fermentation:
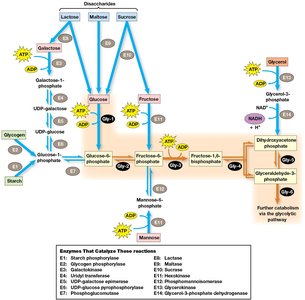
Alternative Substrates for Glycolysis
Cells can utilize various carbohydrates besides glucose, including disaccharides and storage polysaccharides such as starch and glycogen. These are broken down into glucose or glycolytic intermediates.
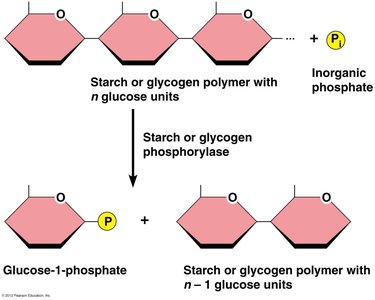
Phosphorolytic Cleavage: Starch and glycogen are cleaved by phosphorylase enzymes to produce glucose-1-phosphate.
Gluconeogenesis: The synthesis of glucose from non-carbohydrate precursors, essentially the reverse of glycolysis.
Regulation of Glycolysis and Gluconeogenesis
Glycolysis and gluconeogenesis are tightly regulated to ensure proper energy balance. Key regulatory enzymes include phosphofructokinase and fructose bisphosphatase, which respond to cellular energy levels and allosteric effectors.
Phosphofructokinase-2 (PFK-2): Regulates levels of fructose-2,6-bisphosphate, a potent activator of glycolysis.
Fructose bisphosphatase-2 (FBPase-2): Regulates gluconeogenesis by removing phosphate groups.
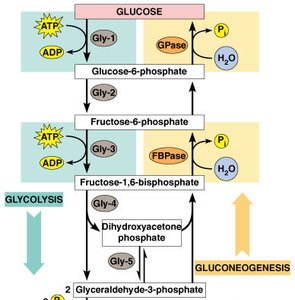
Table: Standard Free Energies of Hydrolysis for Phosphorylated Compounds
This table compares the free energy changes () for hydrolysis of various phosphorylated compounds involved in energy metabolism.
Phosphorylated Compound and Its Hydrolysis Reaction | (kcal/mol) |
|---|---|
Phosphoenolpyruvate (PEP) + H2O → pyruvate + Pi | -14.8 |
1,3-Bisphosphoglycerate + H2O → 3-phosphoglycerate + Pi | -11.8 |
Phosphocreatine + H2O → creatine + Pi | -10.3 |
Adenosine triphosphate (ATP) + H2O → adenosine diphosphate (ADP) + Pi | -7.3 |
Glucose-1-phosphate + H2O → glucose + Pi | -5.0 |
Glucose-6-phosphate + H2O → glucose + Pi | -3.3 |
Glycerol phosphate + H2O → glycerol + Pi | -2.2 |

Additional info: These notes expand on the original content by providing definitions, context, and examples for each major topic, ensuring a comprehensive and self-contained study guide for cell-biology students.
