 Back
BackCh.9: Chemotrophic Energy Metabolism-Glycolysis and Fermentation
Study Guide - Smart Notes

Chemotrophic Energy Metabolism: Glycolysis and Fermentation
Introduction to Chemotrophic Energy Metabolism
Chemotrophic energy metabolism refers to the cellular processes by which organisms extract energy from chemical compounds, primarily through the breakdown of glucose. Glycolysis and fermentation are central pathways in this process, allowing cells to generate ATP under both aerobic and anaerobic conditions.
ATP: The Universal Energy Coupler
Structure and Function of ATP
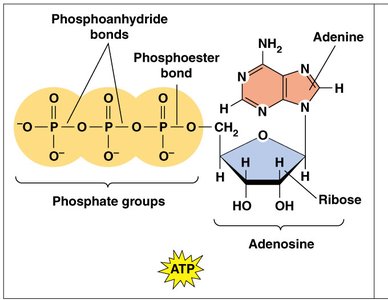
ATP (adenosine triphosphate) is the primary energy currency of the cell. It consists of an adenosine molecule (adenine + ribose) attached to three phosphate groups. The high-energy phosphoanhydride bonds between the phosphate groups are the source of ATP's energy.
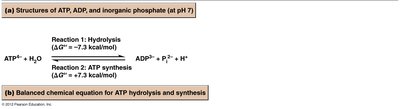
ATP hydrolysis releases energy that can be used for cellular work.
ATP synthesis stores energy by forming new phosphoanhydride bonds.
Standard Free Energies of Hydrolysis
The hydrolysis of phosphorylated compounds releases varying amounts of free energy, measured as ΔG°'. ATP has an intermediate transfer potential, making it ideal for energy coupling in cells.
Phosphorylated Compound and Its Hydrolysis Reaction | ΔG°' (kcal/mol) |
|---|---|
Phosphoenolpyruvate (PEP) | -14.8 |
1,3-Bisphosphoglycerate | -11.8 |
Phosphocreatine | -10.3 |
Adenosine triphosphate (ATP) | -7.3 |
Glucose-1-phosphate | -5.0 |
Glucose-6-phosphate | -3.3 |
Glycerol phosphate | -2.2 |

ATP in Cellular Metabolism
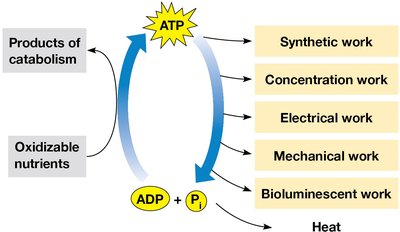
ATP acts as an energy intermediary, linking catabolic (energy-yielding) and anabolic (energy-consuming) processes. Energy from catabolism is used to synthesize ATP, which then powers various cellular activities.
Synthetic work (biosynthesis)
Concentration work (active transport)
Electrical work (nerve impulses)
Mechanical work (muscle contraction)
Bioluminescent work (light production in some organisms)
Redox Reactions in Metabolism
Oxidation and Reduction
Redox reactions involve the transfer of electrons between molecules. Oxidation is the loss of electrons, while reduction is the gain of electrons. These reactions are fundamental to energy extraction from nutrients.
NAD+ (nicotinamide adenine dinucleotide) is a key electron carrier, reduced to NADH during catabolic reactions.
Glycolysis: Processing Glucose to Pyruvate
Overview of Glycolysis
Glycolysis is a ten-step metabolic pathway that converts glucose into pyruvate, generating ATP and NADH. It occurs in the cytoplasm and can function under both aerobic and anaerobic conditions.
Net products per glucose: 2 pyruvate, 2 ATP, 2 NADH
Divided into three phases: Preparation, Cleavage, and ATP Generation
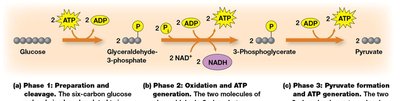
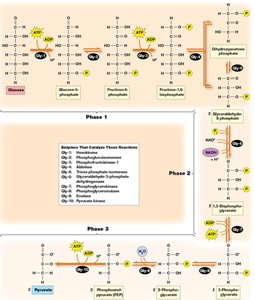
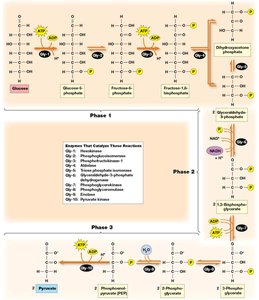
Detailed Steps of Glycolysis
Phase 1: Preparation and Cleavage – Glucose is phosphorylated and split into two three-carbon sugars.
Phase 2: Oxidation and ATP Generation – Glyceraldehyde-3-phosphate is oxidized, producing NADH and ATP.
Phase 3: Pyruvate Formation and ATP Generation – Further ATP is generated as phosphoenolpyruvate is converted to pyruvate.
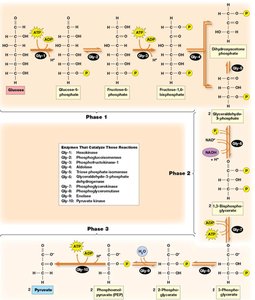
Key Irreversible Steps
Reaction 1: Phosphorylation of glucose to glucose-6-phosphate by hexokinase (irreversible).
Reaction 3: Phosphorylation of fructose-6-phosphate to fructose-1,6-bisphosphate by phosphofructokinase (irreversible, key regulatory step).
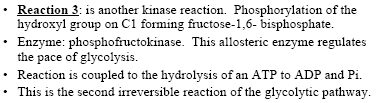
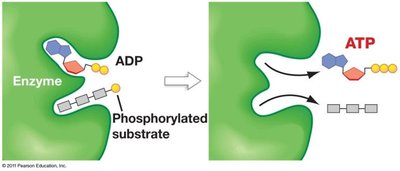
Substrate-Level Phosphorylation
ATP is generated directly in glycolysis by substrate-level phosphorylation, where a phosphate group is transferred from a high-energy intermediate to ADP.
Overall Glycolytic Equation
The net reaction for glycolysis is:
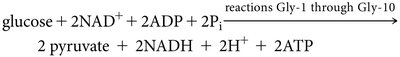
Fates of Pyruvate: Aerobic and Anaerobic Pathways
Aerobic Respiration
In the presence of oxygen, pyruvate is transported into mitochondria and converted to acetyl-CoA, entering the citric acid cycle and oxidative phosphorylation for maximal ATP yield.
Anaerobic Pathways: Fermentation
When oxygen is absent, cells regenerate NAD+ by reducing pyruvate through fermentation. Two main types are:
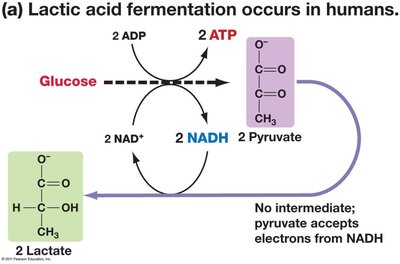
Lactic Acid Fermentation: Pyruvate is reduced to lactate (in animals, e.g., muscle cells during intense exercise).
Alcoholic Fermentation: Pyruvate is converted to ethanol and CO2 (in yeast and some plant cells).
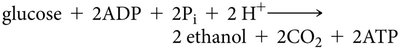
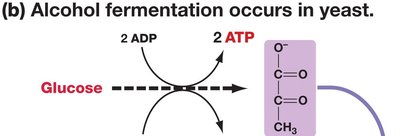
Fermentation Equations
Lactic Acid Fermentation:
Alcoholic Fermentation:
Alternative Substrates for Glycolysis
Entry of Other Sugars
Other carbohydrates such as fructose, galactose, and mannose can enter glycolysis after conversion to intermediates of the pathway. Storage polysaccharides like glycogen (animals) and starch (plants) are broken down to glucose-1-phosphate, which is converted to glucose-6-phosphate for entry into glycolysis.
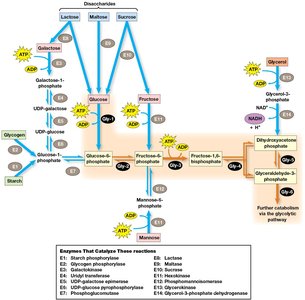
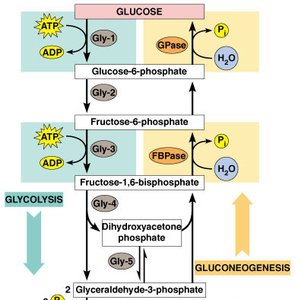
Regulation of Glycolysis and Gluconeogenesis
Key Regulatory Enzymes
Glycolysis and gluconeogenesis are tightly regulated to meet cellular energy demands. The main regulatory enzymes include:
Phosphofructokinase-1 (PFK-1): Major control point for glycolysis, activated by AMP and inhibited by ATP and citrate.
Fructose-2,6-bisphosphate: A potent activator of PFK-1 and inhibitor of fructose-1,6-bisphosphatase (key enzyme in gluconeogenesis).
Summary Table: Comparison of Glycolysis and Fermentation Pathways
Pathway | Oxygen Requirement | End Products | ATP Yield (per glucose) |
|---|---|---|---|
Glycolysis (aerobic) | Oxygen present | Pyruvate | 2 |
Lactic Acid Fermentation | Oxygen absent | Lactate | 2 |
Alcoholic Fermentation | Oxygen absent | Ethanol, CO2 | 2 |
Additional info: The regulation of glycolysis and gluconeogenesis ensures that these pathways do not occur simultaneously in the same cell, preventing a futile cycle and conserving cellular energy.
